Endodontic Topics 2011, 18, 62–77
All rights reserved
2011 © John Wiley & Sons A/S
ENDODONTIC TOPICS 2011
1601-1538
Survival against the odds: microbiology of root canals associated with post-treatment disease
DAVID FIGDOR & KISHOR GULABIVALA
Most periapical lesions resolve after root canal treatment of teeth with primary infections. Over the last decade there has been a renewed focus on post-treatment apical periodontitis and its etiology. This review describes the microbiota associated with persistent post-treatment infection, including microbial identification, ecology, and environmental selection. Compared with untreated teeth, the infection pattern in root canals with post-treatment disease shifts to a resistant, mainly Gram-positive community. The main challenge in root canal re-treatment is access to the residual apical infection. Elimination of the microbial flora, or a dramatic reduction and sufficient ecological shift to allow host tissue healing, remains the biological goal.
Received 28 January 2011; accepted 4 February 2011.
Over millions of years, oral micro-organisms have evolved to co-exist with the mammalian host (1) mainly as biofilms adherent to enamel, dentin, cementum, and the oral mucosa. A dramatic shift in the biofilm ecology is essentially responsible for the development of caries and periodontal disease (2). In contrast, the dental pulp is an intrinsically sterile tissue and apical periodontitis occurs after a breach of the protective enamel/dentin encasement and polymicrobial invasion and infection of the pulp space.
Apical periodontitis is a more widespread disease than moderate or severe marginal periodontitis, affecting 50% of the population by age 50, and 62% of individuals over age 60 (3). Since the prime etiology is infection, clinical management of apical periodontitis is targeted at microbial control by root canal treatment. Many millions of root canal treatments are performed annually (4) and it is estimated that there are many hundreds of millions of root-filled teeth in the adult populations of Western countries (5).
The delivery of sound treatment with successful outcomes is a primary clinical goal. It is natural to expect a higher success rate for root canal treatment when it is performed by trained specialists rather than general practitioners. Based on epidemiological data, the success rate of root canal treatment performed in specialist practice is in the order of 87% compared with 72% for treatment in general practice (3). Although seemingly insignificant, the 15% difference equates to many millions of failed treatments when applied to Western populations (5). In the context of the cost of root canal re-treatment and crown or restoration replacement, the cumulative economic impact is in the order of billions of dollars. Thus, there are broad health, social, and economic consequences of root canal treatment failure and potentially significant benefits individually and collectively if it were possible to reduce the proportion of endodontic failures (5).
Different types of infection
The prime reason for post-treatment disease is infection in the apical part of the root canal system by species that have endured or evaded antimicrobial treatment, survive in the filled root canal, and are capable of inflaming the periapical tissue. Some species are well suited to the barren but protected environment of the filled root canal, yet how microbes take advantage of this unique situation has not been adequately elucidated at this time.
There are two potential pathways for post-treatment infection (Fig. 1). The first is for micro-organisms that were part of the initial polymicrobial consortium of the untreated root canal to prevail and subsequently participate in post-treatment disease. An ability to endure biomechanical preparation is invaluable, but alone is not enough without a capacity to survive in the root-filled canal and inflame the periapical tissue. A second pathway is re-infection of the root-filled canal via microbial leakage around the coronal restoration. It should be noted that this conceptual framework is deduced from currently available information on the composition of the microbial flora in untreated and treated canals, and that direct evidence is lacking to illustrate the natural pathogenesis of the respective infection patterns.
Species that survive treatment
A few studies have provided information on the species that survive instrumentation and antimicrobial irrigation. Generally, Gram-negative species and fastidious anaerobes have a limited capacity to endure treatment and are readily eliminated by instrumentation and antimicrobial irrigation (6). Gram-positive species and facultative anaerobes have a higher rate of recovery in post-instrumentation samples (6–12). It is worth noting that after root canal preparation there are low cell numbers that survive treatment, in contrast to the high initial microbial cell population (7, 13, 14). Any cells surviving treatment are likely to do so in small biofilms and discrete pockets of cells away from the main body of the canal (15, 16). Since there are few surviving cells that are in a vulnerable state, there are implications for sample collection because special methods are necessary for recovery by culture (17). Without these measures, it is highly likely that the sampling procedure will not pick up surviving cells.
Species that survive in the root-filled canal and maintain periapical inflammation
Although some species may elude antimicrobial treatment, if they are left without suitable nutrition and are not in a propitious location, the microbes may not be able to survive in the root canal over time. A number of microbial properties favor the selection and survival of some species over others. For example, an ability to endure starvation is a beneficial survival characteristic which helps ensure that some species outlast others until they may access nutrients from the local milieu, e.g. serum-type fluid that may seep into the canal space over time.
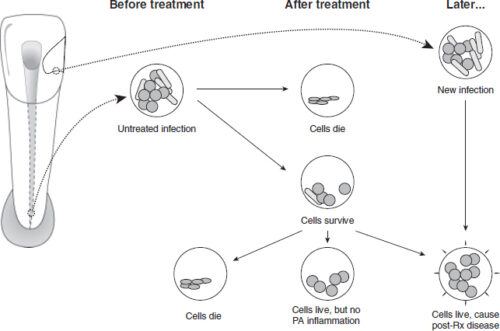
Fig. 1. Potential pathways for post-treatment infection.
Starvation survival characteristics have been demonstrated in selected species known to be involved in persistent infection such as Enterococcus faecalis (18) and Candida albicans (19), whereas others seldom identified in persistent infection have poor starvation survival ability (20).
Although some microbes may weather treatment, their subsequent destiny and involvement in maintenance of the periapical lesion depends on nutritional availability, luck, and location (21). For example, microbes that survive in the isthmus between canals in the mesial root of a lower molar after endodontic treatment (16) may not have the ability to communicate with the periapical tissue and thus not be in a position to inflame host tissue.
Re-infection after treatment
Sound restoration goes hand-in-hand with high quality root canal treatment for an optimal outcome of the treated tooth. Any breach in the marginal integrity of the coronal restoration poses a threat of nutrient or microbial leakage into the root canal system and an ensuing risk to the prognosis. Based on cross-sectional studies, it has been observed that there is a higher incidence of periapical pathoses when there are radiographic signs of poor quality restorations (22–24). While it makes good biological sense that there is a connection between coronal microleakage and the outcome of root canal treatment, some studies point to the root filling as an effective barrier to apical microbial advancement (25). It remains to be shown whether the relationship between poor coronal seal and reduced treatment outcome is a reflection of the (lower) quality of the initial treatment or that coronal leakage was responsible for the subsequent treatment outcome.
Specific versus non-specific microbial etiology
The seminal discovery and publication of Koch’s work on anthrax (26) led to the realization that many classic diseases are the result of individual pathogens with a specific microbial etiology. Yet the insightful microscope and culture observations of Miller in 1894 (27) showed that apical periodontitis is associated with a collection of species in a polymicrobial infection. That the infection is polymicrobial means that it is difficult to attribute specific roles to individual species. Thus, all species present and living have the potential to contribute to the infection process (21) and, together, they may act collectively for the benefit of the community (28). Factors such as strain variation may also account for variable pathogenicity (29–31).
The fundamental reason for accurate and complete identification of bacterial communities from root canal systems is to disclose those species or species-combinations that may play key roles in the progress of the disease or its acute exacerbation, especially those that may be resistant to conventional therapy or implicated in treatment failure (32, 33).
The association between the type of root canal flora or individual species and periapical lesion development has been studied by several groups of investigators. Classic studies on the periapical responses to indigenous bacterial infections in the monkey model used an experimental design in line with Koch’s postulates for a polymicrobial infection (34–36). Teeth that were infected by exposure to the oral cavity developed periapical lesions; the proportion of facultative anaerobic species decreased and strict anaerobic species increased over a 6–7 month period (36). There are changes in the type and proportions of the flora over time as well as location within the root canal, as shown by animal experiments in monkey (35), dog (37), and rat teeth (38), which induce species-specific host responses (39, 40).
Species work together to support growth and survival and co-operate to thwart the host defense. Other strategies may include gene transfer within the biofilm matrix for the purpose of enhancing virulence or evading the host response (41, 42). Periapical lesion development therefore seems to be dependent upon the nature of the mixed infection, the succession within it, and its ultimate survival. Some species, however, are capable of survival and host stimulation as sole pathogens (34, 43).
The clinical spectrum of apical periodontitis varies from acute to chronic with suppurative presentations and the range of responses may be a function of variation in host response (44) and/or a function of fundamentally different types of microbial flora (40). Different pathogenic profiles within the microbiota raise the prospect of different susceptibilities to treatment and the prospect of specific treatment protocols.
Biofilm ecology
Medical microbiology has been founded on treating infections conceptualized as individual planktonic cells colonizing tissue surfaces and compartments. It has become apparent that this perception of independent single cells, which survive by moving towards nutrients and away from harm, is actually not the preferred mode of bacterial survival. The natural pattern is a sedentary existence in enclosed extracellular matrix communities that are frequently multi-species in composition and adherent to surfaces. This is a basic description of biofilms, which were first recognized in root canal systems by Nair (45). Although the term “biofilm” was not used, Nair (45) described the presence of clusters of cells thus:
Most of the flora in the root apex remained suspended in an apparently moist canal lumen. Less frequently, dense aggregates of bacteria could be observed sticking to the dentinal wall of the root canal. The interbacterial spaces revealed an amorphous material, particularly towards the center of the aggregates. The single or multi-layered bacterial condensations were visible only at EM level.
Subsequently, he revised this interpretation to suggest that the majority of the bacteria were probably embedded in a surface adherent biofilm (personal communication), a view confirmed in other studies (46). The study of endodontic biofilms is in its infancy, but it is fortunate that the process of biofilm formation follows a universal pattern and Endodontics can borrow many established concepts (47).
The survival attributes of a motile single cell are dramatically different from those required for sedentary community life. Precisely what surface conditions encourage colonization on root canal dentin remains to be determined (48), but colonizing species act together to optimize the utilization of local resources (43, 49). Such sophisticated bacterial collaboration requires intercellular communication. Cells release specific signal molecules into the environment and, through diffusion or convective distribution and detection by other cells, they communicate when they have reached a critical cell density (50–52). In this way, the collective population co-ordinates efforts in nutritional utilization, virulence expression, and various other community functions. Thus, the extracellular matrix develops as a co-ordinated affair, allowing fluids to circulate and an exchange of cellular components and nutrients amongst the microbial community (53, 54).
Some cells play an important role in the survival of the community. Persister cells neither grow nor die (55) and are thought to have some responsibility for resistance to antimicrobials (56). Biofilms may also enhance their chances of survival by dispersing breakaway portions or individual cells in a planktonic phenotype (57). It is therefore possible to visualize localized variations in biofilm composition in the root canal system, for which some evidence exists (46, 58, 59).
In summary, biofilms are highly organized multicellular, multi-species structures that collaborate collectively to harvest nutrients, and display sensing and survival tactics. There is a need for more sophisticated strategies to control biofilms, not only in chemomechanical terms but especially in biological terms (53, 60). In the untreated infected root canal, there is a complex and rich biofilm community, but biomechanical instrumentation results in a dramatic change to the environment. Root canal treatment causes massive disruption and dissolution of the biofilm and strips away most of the nutrient supply. In cases treated in accordance with accepted principles, only small pockets of bacterial communities may survive in limited spaces of the root-filled canal (16). How long they survive after root filling and whether they evoke post-treatment disease is currently unknown.
Microbiology of untreated root canal infection
The hard and soft tissues of the oral cavity are home to a rich and diverse assortment of micro-organisms, previously estimated with culture methods at >4500 different kinds (61), and with molecular techniques and re-classification this number has more recently been revised upwards to >700 species (62). Each individual is estimated to have 100–200 species of oral bacteria (63) and all have the potential to invade the pulp space, yet a relatively small number of species are typically recovered in root canal samples. Using advanced culture techniques, a limited assortment of species has been reported (33, 64–66), typically a polymicrobial mix of 2–12 species.
Molecular methods have the potential to recover DNA from difficult-to-culture species and, while a broader range of species-specific DNA has been described (28, 67, 68), the number and diversity of species in individual root canals is typically 10–30 species (28, 68), which remains well below that reported in the oral cavity (69) or identified in the periodontal pocket (63).
Strong selection pressures define the type and course of infection. The local environment primarily governs microbial composition and organization, and the most important factors are anaerobiosis, microbial interactions and nutrition. Obligate anaerobes dominate the microbial flora in root canal infections of human teeth with intact pulp chambers (33, 64–66). Several studies have shown that particular species have a tendency to associate together (70–72), and there is good reason for microbes to co-operate collectively so that they can flourish in a communal environment. Individual species may supply essential nutrients for the growth of other members of the population (73–75). Bacteria may also compete for nutrition, or inhibit the growth of others by production of bacteriocins (76).
Nutrition is essential for microbial growth – nutrients may potentially be derived from the oral cavity, connective tissue and blood components in degenerating pulp (77), dentinal tubule contents, or a serum-like fluid from periapical tissue (78).
Thus, apical periodontitis is caused by a habitat-adapted polymicrobial infection of the pulp space. The microbial flora typically consist of a restricted group of species, with equal proportions of Gram-positive and Gram-negative species, dominated by anaerobes with fastidious environmental and nutritional requirements (33, 75).
Ecology and species
The root canal infection is a dynamic process and various bacterial species dominate at different stages of this process. In a long-standing infection, there is a shift towards dominance of the community by selected species. The most important factors driving this development are availability of nutrition, oxygen level (redox potential), and the local pH within the root canal (21, 33).
Exogenous nutrients, such as fermentable carbohydrates, can affect the microbial ecology of the coronal parts of an exposed root canal, but endogenous proteins and glycoproteins are the principal nutrients in the main body of the root canal system (21, 79, 80). Although there is a restricted supply of proteins in the root canal from progressive degradation of the small volume of pulpal tissue, the bacteria induce a periapical inflammation that leads to influx of a serum-like exudate into the canal. This fluid is a sustainable nutrient source containing proteins and glycoproteins for those bacteria that have a proteolytic capacity. The bacteria which dominate this stage of the infection are likely to be those that either have proteolytic capability, or maintain a co-operative synergy with those that can utilize this substrate for bacterial metabolism (21, 79, 80). Bacterial metabolism of the serum-like fluid also causes reduction of the redox potential and a concomitant rise in the pH (2).
Differences between culture and molecular results
Elaboration of the root canal microbial flora was established using sophisticated anaerobic culture methods that were specifically developed for growing many obligately anaerobic human pathogens (81). Advances in molecular techniques have increased our ability to differentiate bacteria and led to the establishment of new genera and species. Many of the taxonomic refinements have developed as split-offs from previously established genera and species. Because classical culture and molecular methods both have distinct advantages and drawbacks, it makes good sense to recognize their respective attributes in order to gain the most from studies on root canal microflora. Successful cultivation relies on viable micro-organisms that live and grow on plates or in liquid media. Molecular approaches, specifically PCR, depend on isolation and amplification of target-specific DNA, regardless of cell viability. With both approaches, the sample is obtained in the same way by soaking fluid from the root canal onto paper points and storing in transport medium. Thereafter, processing differs in the laboratory. Some of the respective features are summarized in Tables 1 and 2.
Conventional bacterial identification has been based on an array of biochemical tests, which are particularly technique- and operator-sensitive. Relatively minor differences in culture procedures can influence results and some characteristics may not be reproducible under conditions used in different laboratories. In addition, the biochemical tests are greatly influenced by the size and age of the inoculum and the degree of anaerobiosis (33). In early studies, isolates were identified only to the genus level. By 1974, the first thorough bacteriological analysis to the species level was published (64), which was subsequently followed by others who adopted more comprehensive procedures for identification (65, 66, 82).
Bergey’s Manual of Systematic Bacteriology (83) was used as the early standard for identification (32, 81, 84, 85). Later, others (65, 66, 82) adopted the VPI manual (86) for anaerobic bacteria. The introduction of biochemical test kits simplified laboratory procedures (87) and from the 1980s the API kits became popular (88). These offered a degree of standardization across laboratories (89) and were often supplemented with other tests (Bergey’s and the VPI manual) as their databases were unable to identify all endodontic isolates (89–92). More recently, commercially available kits have been used exclusively with identification based entirely on the kit database (93–97). This is problematic because the databases do not cover the strains found in root canal systems (98).
Table 1. Advantages and disadvantages of culture-based methods
| Advantages | Disadvantages |
| Assesses living (culturable) microbes | Risk of contamination |
| Able to recognize viable cells in a sample | High skill level is necessary for optimal results |
| Easy to quantitate cells in a sample | Time and resource intensive |
| High sensitivity with appropriate media | Relies on phenotypic biochemical characterization |
The use of molecular techniques for identification of bacteria in Endodontics is relatively recent (99). The studies have used the comparative 16S rRNA gene sequence approach to identify bacteria in root canal samples. The majority have applied species-specific PCR amplification of variable 16S rRNA sequences for identification. Some of the target species include Actinomecetales (99), Fusobacterium (99–102), Bacteroides forsythus (99–101, 103–105), Streptococcus species (99, 102, 104), Prevotella intermedia and nigrescens and black pigmented species (106, 107), Treponema (105, 108–110), Slackia exigua, Mogibacterium timidum and Eubacterium saphenum (111), Prevotella spp. (101), Porphyromonas spp. (101, 105, 109), Peptostreptococcus spp. (101), and Enterococcus faecalis (101, 112).
Some studies have used DNA hybridization probes to detect specific bacteria in samples (100, 103, 104, 113). Few of these studies have conducted sensitivity/specificity checks on their DNA probes (111) and few have sequenced amplicons to confirm identity (67, 68, 99, 109, 111, 112, 114). Only a few studies (67, 68) attempted to carry out a microbial community analysis using both cultivation and molecular cloning approaches.
The influence of various factors in sampling, detection, and identification method on species recovery was shown in a study that combined both methods (115). A clinically intact tooth with a periapical radiolucency, extracted for restorative reasons, was immediately placed in an anaerobic chamber and surface-decontaminated; the pulp chamber was accessed and the root canal was conventionally sampled with paper points. The tooth was then cryo-pulverized for the second sample. Both samples were analyzed by culture-dependent and culture-independent molecular techniques (16S rDNA amplification, cloning, sequencing). Samples of the tooth yielded 44 taxa; 24 in the root canal and 28 in the tooth, but only 8 were common to both samples. By culture, 23 taxa were identified and 27 by PCR cloning, but only 5 were common to both methods. The culture method revealed 16 bacterial species in the root canal compared with 11 in the tooth, but only 4 were common to both samples. This simple experiment illustrates the potential for sampling and detection methods to introduce distortions in identification of the root canal flora.
Table 2. Advantages and disadvantages of molecular-based methods
| Advantages | Disadvantages |
| Assesses DNA sequences (target-specific with PCR) | Risk of contamination |
| Potential to recover majority of microbes | Relies on established sequence identities |
| Modest skill level required | Relies on well-designed primer set |
| Relatively quick and straightforward processing | DNA recovery influenced by cell lysis method |
| Can freeze samples for later processing | Unable to distinguish live from dead cells |
| High specificity |
Why is there a disparity between the methods?
Historically, one of the most important challenges in determining microbial diversity has been that microorganisms are extracted from their environment and then studied microscopically, or in culture. There is clear evidence that not all organisms present in the root canals are subsequently cultivated, as revealed by microscopy of direct smears (27, 32, 85, 116) and by correlative light and electron microscopy (45).
The diversity of microbial communities is usually based on complex environmental, nutritional, and communicative interactions between species. By necessity, the process of taking a sample underestimates both the number and variety of species present in the original ecological niche (117, 118). The sampling method, conditions of transport, storage, culture (dilution, handling, media, incubation and atmosphere), and laboratory sub-culture all have the potential to bias the resulting types and numbers of species (119). At sampling, bacteria may be dormant, fragile, or in a starvation state, which may also influence recovery (120, 121).
Recovery and analysis of nucleotide sequences in a biological sample is completely independent of microbial viability or growth. By recovering available DNA, the molecular approach has an advantage over conventional culture because there is the potential to detect all culturable, previously uncultured, and not-yet-culturable micro-organisms in mixed samples (122). These methods also have drawbacks as each physical, chemical, and biological step may bias sequence recovery (123, 124). Whether the sample sequences are representative depends on the process of sample collection, transport, and efficacy of the DNA extraction. Successful isolation of DNA is influenced by physico-chemical factors in the sampled environment (e.g. inhibitors and nucleases) and biological properties of individual species (e.g. Gram-negative species are more susceptible than Gram-positive species to cell lysis) (123, 124). Some factors that influence the molecular analysis of the microbial flora in mixed samples are summarized in Table 3.
It should be pointed out that there are few studies which have evaluated samples from the same case by both culture and molecular approaches (67, 68, 125). In all such studies there is a disparity between the culture and molecular findings, where some species are identified by both approaches and some species are detected by one but not the other method. More work is needed in this area to satisfactorily reconcile, clarify, and resolve the reasons for different results from these two approaches.
Micro-organisms involved in post-treatment disease
The goal of instrumentation and irrigation of the root canal system is infection control. However, limitations in access to micro-organisms and in treatment efficacy mean that conventional root canal treatment falls short of this goal and low numbers of bacteria may survive in 30–50% of cases (7, 13, 14, 134–138). Application of an inter-appointment antibacterial dressing in multi-visit treatment does improve bactericidal efficacy (134, 137, 139–141) before canal filling. The root filling can entomb residual bacteria and deprive them of nutrients, and block direct communication with the periapical tissues. Persistent periapical disease requires that residual bacteria maintain communication with the periapical tissues and are capable of eliciting inflammation. Therefore, a distinction is made between species that survive root canal treatment and species involved in post-treatment disease (Fig. 1).
Table 3. Some factors that influence molecular analysis of micro-organisms in mixed samples
| Co-extraction of PCR inhibitors (humic acids, polysaccharides) with nucleic acids (124) |
| Changes in composition of 16S rDNA clone libraries caused by environmental DNA concentration (126) |
| G+C composition of 16S rRNA genes (127) |
| Genome size and copy number of rRNA genes (128, 129) |
| Lack of information on genome size and gene copy number for uncultured micro-organisms (124) |
| Choice of PCR primers, their design, and number of replication cycles (130) |
| Differential cloning efficiencies of PCR amplicons (123) |
| Formation of mosaic sequences from separate genes (131) |
| Limitations in quality and integrity of sequence databases (132, 133) |
Many studies have described bacteria present in post-treatment samples and there is a clear tendency for Gram-positive species to prevail after antimicrobial endodontic treatment (7, 9, 11, 12, 142). While there are no particular definitive resistant micro-organisms, certain species are reported in greater frequency as survivors after instrumentation, in particular but not exclusively, Streptococcus, Propionibacterium, Eubacterium and Actinomyces species (9, 143).
Will all species live and maintain a periapical inflammation?
Some bacteria may survive chemomechanical treatment but do not have the capacity to induce or maintain apical periodontitis. Based on culture studies of samples taken after instrumentation and irrigation, there are no recoverable bacteria in 50–70% of canals (7, 134–136, 138, 143). The converse of this is that 30–50% of cases will have recoverable bacteria at the time of root filling, yet not all of these cases end in post-treatment disease. In one study, recoverable bacteria were present in 40% of teeth at the time of root filling, yet in those cases with infection, 68% healed (9). This demonstrates that the presence of infection at the time of root filling does not always result in post-treatment disease. This was confirmed histologically in a study evaluating the impact of contemporary root canal treatment procedures on the microbiota in the mesial roots of mandibular molars with periapical lesions; it was found that 14/16 roots still demonstrated residual biofilms in the apical complexities after single-visit treatment (16).
Since the success rate of single-visit root canal treatment is in the range of 70–85%, it raises the question of the fate of the residual microbiota in cases that heal. Entombed in the filled canal, they may die, but in some cases residual bacteria may continue to interact with the host tissues, resulting in a slow healing response over many years (144–146). In other cases, it is reasonable to think that some species endure the low nutrient or starvation conditions and survive in the filled root canal, which is consistent with a less favorable healing outcome in cases where infection is present at the time of canal filling (9, 147). Without access to the periapical tissue, the lesion would likely heal; however, there remains a risk of persistent apical periodontitis if microbes survive and have an opportunity to communicate with the periapical tissues.
Intraradicular infection
In untreated cases, analysis of the root canal infection depends on careful, contamination-free access to sample the microbiota, which typically contain high cell numbers of multiple species. In studies of infected root-filled teeth, a significant challenge is retrieval of the microbial flora, usually in low numbers, from the filled root canal system. Removal of the root filling by mechanical instrumentation may generate heat and the use of solvents (usually chloroform) may kill remaining bacteria. One study compared the impact of using chloroform on the recovery of bacteria and found that 78% of the canals were positive for bacteria without chloroform while only 48% were positive with its use (148). Some studies specifically exclude the use of chloroform before microbiological examination for this reason (149–153).
In general, the bacterial species recovered from root-filled teeth are mostly a subset of those found in untreated teeth, but with a reduced species diversity and quantity. Typically, Gram-positive facultative bacteria dominate the microbiota (81, 148, 153). In poorly treated teeth, the microbial flora resemble those recovered from untreated teeth (149, 153–156).
A consistent finding across multiple studies is the frequent isolation of E. faecalis (81, 148, 150–154, 156–160), although some studies failed to identify it (67, 149) and some studies describe the presence of E. faecalis in a small proportion of cases (161, 162). The presence of E. faecalis is notable not only because of a high prevalence in previously root-filled teeth, but it is usually infrequently found in teeth with primary infection (7, 72, 163). This apparent paradox has raised questions about how E. faecalis has come to be present in a high proportion of teeth after, but not before, root filling (based on cross-sectional studies) (21, 164). One likely contributory factor is the operative events during the period of the original treatment. E. faecalis has been reported in a significantly higher proportion of cases when there has been protracted treatment or the canals have been left open (165). Another question is whether the source is resident in the host oral microflora or is of transient derivation, and a recent study points to an exogenous source of E. faecalis (166). When present in teeth with post-treatment disease, it is more difficult to eradicate during subsequent re-treatment (153).
Other species that appear with greater prevalence in root-filled teeth with post-treatment disease include Streptococcus species (148–150, 152–154), Candida species (predominantly C. albicans) (148–150, 152–154, 156, 167, 168), and Propionibacterium species (169, 170). In culture-independent studies, there are reports of Filifactor alocis and Dialister species (156, 160, 170, 171).
In most studies, there is a reduced diversity of isolated species when compared to the microbiota of untreated teeth. In culture-based studies, there is a higher incidence of monoinfections, particularly of E. faecalis (148, 150, 151, 153), although molecular-based studies report a greater number of species (170, 172).
Extraradicular infection
The periapical granuloma is in intimate anatomical contact with the root apex and frequently communicates with the apical root canal, so obtaining a bacteriological sample from the periapical tissues without contamination from either the root apex or canal presents an intricate challenge. It is also the source of controversy in this area. Almost 50 years ago Möller demonstrated that, with a conventional surgical approach using routine isolation and suction, it was impossible to recover periapical tissue without contamination (81). Contamination control samples from adjacent soft tissue and bone were repeatedly positive, which invalidated the targeted periapical tissue sample. He solved the problem by stringent isolation using a custom-made acrylic shield, which allowed him to work and take samples without contamination from the surrounding field (81). It is important to recognize the significance of this accomplishment because bacteriological studies using root-end scrapings or samples including the root tip, surgical samples without stringent isolation, and periapical tissue sampling via the root canal are subject to an unacceptable risk of contamination. If the overlying soft tissue is carefully isolated, cleaned, and disinfected, it is possible to successfully take samples by needle aspiration of the periapical granuloma.
The interaction of root canal microbes with the host occurs in a dynamic interplay at the root apex. Small groups of bacteria may breach the apical foramen and the host’s normal immune response is to detect and destroy invading bacteria. This is the role and function of the periapical granuloma. In some cases, bacteria may interfere with the host defense and overcome it, which is the reason for the presention of acute symptoms such as pain, swelling, or exudation. The vast majority of bacteria succumb to the host response, occasionally with adjunctive clinical support in acute cases, and are therefore not generally considered a sustainable form of extraradicular infection.
Only a few oral species have a demonstrated capacity for establishment and long-term survival in host tissue, independent of the root canal system. Verification that the infection is extraradicular and unconnected with the root canal system in such cases is borne out by correlative light and electron microscopy of serial sections, illustrated by a tooth with a periapical actinomycosis (173). This need not exclude some cases where there may be a contiguous arrangement between Actinomyces species in the granuloma and apical root canal. The clinical significance of the extraradicular location is that conventional root canal treatment will not address the source of the persisting infection. Periapical surgery is required to remove it.
The main genera involved in extraradicular infections are Actinomyces species and Propionibacterium species (174–176). The presence of A. israelii is a recurrent finding in therapy-resistant cases (72, 163, 176) and is the most common species involved in actinomycosis (177). Of the Actinomyces species, A. israelii is isolated at twice the prevalence of Actinomyces gerencseriae (formerly A. israelii serotype II) in human abscesses (177) but in half the prevalence in primary root canal infections (163). The role of A. gerencseriae in persistent infection after root filling remains to be clarified. Another Actinomyces species, Actinomyces radicidentis, is occasionally associated with post-treatment disease (169, 178).
Environmental selection
Lourens Baas Becking, a Dutch microbiologist, is credited with the statement “Everything is everywhere, but the environment selects,” which refers to the local environment as a critical determinant in microbial ecology and survival. In the oral cavity, distinct microenvironments at various soft and hard tissue surfaces influence the composition of the microbiota. The untreated infected root canal system is an environment that provides micro-organisms with a rich source of nutrition. Initially, there may be a source of carbohydrates facilitating growth of facultative anaerobes, but as the infection develops over time, the nutrients are mainly peptides and amino acids, which favor anaerobic proteolytic species.
In the filled root canal, most of the nutrients have been stripped away during earlier chemomechanical cleaning, leaving comparatively barren conditions for surviving microbial cells. These microbes generally face a low-nutrient or starvation environment, but in favorable conditions may encounter a serum-like fluid transudate from the periapical tissue.
Some species and individual strains are better equipped to endure periods of starvation, which enhances their long-term survival capacity. Species that are more prevalent in post-treatment disease, such as E. faecalis and C. albicans, have been shown to have a superior starvation survival capacity (18, 19) compared with strict anaerobes that dominate the microbiota in untreated cases (20), which helps explain why the latter group rarely participates in post-treatment disease.
In the extraradicular environment, cellular and humoral factors are deployed by the host to routinely eliminate micro-organisms that invade the periapical tissue. An ability of the micro-organism to strategically respond to the host defense is crucial for survival. The microbial arsenal includes evasion by physical concealment or biological evasion of host surveillance, cell-mediated phagocytosis, or evasion of immune protein-mediated antibodies and complement. Species involved with extraradicular infection, such as A. israelii and A. radicidentis, have been shown to survive in host tissue in animal experiments (179–183) through bacterial cohesion and avoidance of phagocytosis.
There are many further factors that constitute the microenvironment such as the host surface and local defense, microanatomy, anaerobiosis, pH, etc. Much more work is needed in this area and discovery of specific environmental factors that regulate local microbial ecology should open up therapeutic options which are more precisely directed at particular types of biofilms and sites.
Conclusion
The effectiveness of root canal treatment depends on microbial control through a combination of direct bactericidal and ecologic effects. Hardier species that have a greater capacity to resist antimicrobial treatment and that adapt to a nutrient-depleted environment are best suited to endure in the root-filled canal. Where microbes survive, facultative Gram-positive bacteria dominate the remaining bacterial communities.
Post-treatment disease is primarily due to persisting (or possibly recurrent) infection of the apical part of the root canal system. An ineffective first attempt at root canal treatment may leave a microbiota that is more difficult to eradicate and located in anatomically complex regions of the root canal system.
The re-treatment strategy for post-treatment disease may depend on the case type. In teeth with inadequate previous treatment, such as missed canals, the infection resembles an untreated case and it should respond well to conventional root canal treatment. In cases of sub-standard treatment, the infection is likely dominated by Gram-positive species, which may be more resistant to re-treatment. In well-treated teeth, post-treatment disease may be due to persistent intra- or extraradicular infection and, in addition to a conventional chemomechanical approach, consideration should be given to surgical intervention. The treatment priority remains the same in all cases: to obtain access to the apical root canal anatomy and control residual infection.
Healing outcome data indicates that around 85% of periapical lesions resolve after root canal treatment of teeth with primary infections. In cases requiring re-treatment, about 75% of periapical lesions resolve over time. Taken collectively, conventional root canal treatment offers an excellent outcome in terms of the periapical healing rate. Nevertheless, many important questions remain. Why some microbes are able to participate in post-treatment disease in some cases is still not well understood. The contribution of microbial dynamics and pathogenicity, specific anatomical attributes, and the role of the host defense are important factors that await clarification.
Over the past two decades, there have been many advances in instrumentation techniques, materials, and technology. The widespread adoption of the operating microscope has meant that the clinician can see the task with far greater visual acuity. Future improvements in root canal treatment will likely come from a deeper biological insight into the microbial pathogenicity and the factors regulating community behavior.