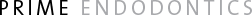A big role for the very small — understanding the endodontic microbial flora
Abstract
Apical periodontitis, an inflammatory process around the apex of a tooth root, is primarily a sequel to microbial infection of the pulp space. The microbial flora is composed of a restricted group of the total oral flora, selected by environmental pressures of anaerobiosis, nutrition and competition with other species and inhabits the root canal as a biofilm of coaggregated communities in an extracellular matrix. The untreated infected canal is generally composed of a polymicrobial mix with approximately equal proportions of Gram-positive and Gram-negative species, dominated by obligate anaerobes.
The type of microbial flora in the root-filled tooth with persistent apical periodontitis has very different characteristics. These infections are characterized by one or just a few species, predominantly Gram-positive micro-organisms with an equal distribution of facultative and obligate anaerobes. Enterococcus faecalis has been a conspicuous finding in most studies.
Because the primary aetiological problem is infection, endodontic treatment is directed at control and elimination of the root canal flora by working in a sterile way. Based on current knowledge, the best available method for obtaining clean, microbe-free root canals is by instrumentation with antimicrobial irrigation reinforced by an intracanal dressing with calcium hydroxide.
Key words: Root canal flora, ecological niche, antibacterial treatment, root canal preparation, root canal irrigants.
Abbreviations and acronyms: EDTA = ethylenediamine tetra-acetic acid; FTM = fluid thioglycolate medium; NaOCI = sodium hypochlorite solution; SEM = scanning electron microscope.
INTRODUCTION
Of the major dental diseases, infection of the root canal is unique for the oral cavity since infection establishes where micro-organisms have not previously been present. The other microbial diseases of the oral cavity, caries and periodontal disease, develop at sites where a microbial biofilm is already established and disease occurs with a change in the environmental conditions, the type and mix of microbial flora.1
In the oral cavity, there are an estimated 1010 bacteria2 consisting of more than 500 different kinds of micro-organisms3,4 and all seek a niche and nutrition. As long as the enamel and cementum layers are intact, the pulp and root canal are protected from invasion, but loss of these structures by caries, cracks or trauma opens an avenue for penetration of bacteria through the dentinal tubules. Leaving behind the nutritionally rich and diverse environment of the oral cavity, microorganisms that establish in the root canal must breach enamel, invade dentine, overwhelm the immune response of the pulp and settle in the remaining necrotic tissue.
Bacteria are everywhere, but the environment selects
All bacteria within the oral cavity share the same opportunities for invading the root canal space, however only a restricted group of species have been identified in infected root canals.5-8 The reason for the disproportionate ratio between potential and actual number of species is that the root canal is a unique environment where biological selection drives the type and course of infection. An anaerobic milieu, interactions between microbial factors and the availability of nutrition are principal factors that define the composition of the microbial flora.
In the initial phase of a root canal infection, the number of species is usually low. If the way of invasion is via caries, the bacteria in front of the carious process are the first to reach the pulp. In cases where there is no apparent communication with the oral cavity and the bacteria penetrate through dentinal tubules, as in trauma cases without pulp exposure, there is no clear pattern of primary bacterial invaders.7,8 The number of bacterial species in an infected root canal may vary from one to more than 12, and the number of bacterial cells varies from <102 to >108 per sample. A correlation seems to exist between the size of the periapical lesion and the number of bacterial species and cells in the root canal. Teeth with long-standing infections and large lesions usually harbour more bacterial species and have a higher density of bacteria in their root canals than teeth with small lesions.
Studies on the dynamics of root canal infections have shown that the relative proportions of anaerobic microorganisms and bacterial cells increase with time and that the facultatively anaerobic bacteria are outnumbered when the canals have been infected for three months or more.9 The endodontic milieu is a selective habitat that supports the development of specific proportions of the anaerobic microflora. Oxygen and oxygen products play an important role as ecological determinants in the development of specific proportions of the root canal microflora.10-12 The consumption of oxygen and production of carbon dioxide and hydrogen along with the development of a low reduction-oxidation potential by the pioneer species favour the growth of anaerobic bacteria.
Nutrition as an ecological driver
The type and availability of nutrients is important in establishing microbial growth. Nutrients may be derived from the oral cavity, degenerating connective tissue, dentinal tubule contents, or a serum-like fluid from periapical tissue. Exogenous nutrients, such as fermentable carbohydrates, affect the microbial ecology of the coronal part of an exposed root canal by promoting growth of species that primarily obtain energy by carbohydrate fermentation. Endogenous proteins and glycoproteins are the principal nutrients in the main body of the root canal system and this substrate encourages the growth of anaerobic bacteria capable of fermenting amino acids and peptides.
The succession of strict over facultative anaerobes with time is most likely due to changes in available nutrition, as well as a decrease in oxygen availability.9 Facultatively anaerobic bacteria dominated by streptococci grow well in anaerobiosis, however their prime energy source is carbohydrates. A decrease in availability of carbohydrates in the root canal occurs when there is no direct communication with the oral cavity, which severely limits growth opportunities for facultative anaerobes.
Growth of mixed bacterial populations may depend on a food chain in which the metabolism of one species supplies essential nutrients for the growth of other members of the population.13-17 Black-pigmented anaerobic rods (Prevotella and Porphyromonas species) are examples of bacteria that have very specific nutritional requirements. They are dependent on vitamin K and hemin for growth. Vitamin K can be produced by other bacteria.18 Hemin becomes available when haemoglobin is broken down, but some bacteria may also produce hemin. A wide range of nutritional interactions is recognized among oral bacteria and these may also influence the associations between bacteria in the root canal.19-21
After degradation of pulp tissue, a sustainable source of proteins develops because bacteria induce periapical inflammation that leads to an influx of a serum-like exudate into the canal. This fluid contains proteins and glycoproteins, and the bacteria that dominate this stage of the infection are likely to be those that either have aproteolytic capacity, or maintain a cooperative synergy with those that can utilize this substrate for bacterial metabolism.
Flora in untreated root canals
The species commonly recovered by culture from root canals of teeth with apical periodontitis have been described in a previous review.8 Because the root canal environment and nutritional supply govern the dynamics of the microbial flora, it means that the bacteria present in the root canal will depend on the stage of the infection.
Initially, there may be no clear associations between bacteria, but strong positive associations develop among a restricted group of the oral flora due to the type of nutrients in the environment.20,22-24 Thus, F. nucleatum is associated with P. micros, P. endodontalis and C. rectus.20 Strong positive associations exist between P. intermedia and P. micros20,24 and Peptostreptococcus anaerobius.20 There is also a positive association between P. intermedia, and P. micros, P. anaerobius and the eubacteria.20 In general, species of Eubacteria, Prevotella and Peptostreptococcus are positively associated with one another in endodontic samples.20,22,24 Properties that these bacteria have in common are that they ferment peptides and amino acids and are anaerobic,25 which indicates that the main source of nutrition in root canals are tissue remnants and a serum-like substrate.
Bacteria in a root canal infection do not occur in vivo as separate colonies, but grow within an extracellular matrix in interconnected communities as a bacterial biofilm. An accurate depiction of the ultrastructural appearance of these biofilms in the infected root canal was first reported by Nair, who described them as co-aggregating communities with a palisade structure.26 The clinical significance of a biofilm growth pattern is that bacteria are relatively protected within the co-aggregated community compared with planktonic forms and are known to be more resistant to antimicrobial treatment measures.27-29 Currently, limited information is available on the development, physiology and antimicrobial management of biofilms in the root canal, however this area should provide a fruitful subject for future research.
Contribution of molecular techniques
An improved systematic structure has been made possible with the application of molecular tools to obtain data from 16S rRNA gene sequences,30 which allows enhanced differentiation between microorganisms and led to the establishment of new genera and species. During the last decade, molecular techniques have been used for microbial identification of root canal samples. Many of the species that are reported as new are split off from previously established genera and species, but the ease of identifying culture-difficult species and the specificity of PCR-based methods has meant that some additional species can be included as typical of the microbial flora of the infected root canal. These include spirochaetes,31-35 and the species Tanerella forsythensis (formerly Bacteroides forsythus),36-40 which are prevalent in infected root canals yet difficult to cultivate.
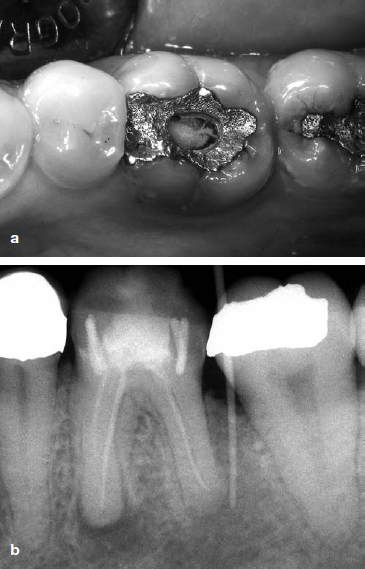
Fig 1. Examples of procedural errors during treatment that lead to failure. (A) Case 1, poor access cavity design, open access cavity and breakdown of the amalgam restoration. (B) Case 2, missed canal and poorly instrumented and obturated canals.
Whilst molecular methods greatly facilitate identification of culture-difficult species and enhance the precision of taxonomic grouping, it is important to recognize the limits as well as the contributions of PCR-based methodology. The high sensitivity of this method implies that it is essential that contamination controls be strictly applied, as contaminants may be easily picked up in the sample and amplified by PCR. The PCR technique is based on recognition of gene sequences — not recovery of cultivable cells capable of growth — so the main drawback of PCR-based methods is that it may detect both living and dead bacteria. Because DNA that persists after cell death may be detected by PCR, the findings from root canal samples may reveal more than just active contributors but could also reflect a historical record of the microorganisms that have entered and not survived in the root canal. Culture-based methods also have their limitations, which include a high degree of skill, labour and time for identification of species and that some species are culture difficult or impossible to culture in vitro. These issues are discussed more fully elsewhere,41 but it is fair to say that both culture and molecular methods each specifically contribute to the study of the root canal flora.
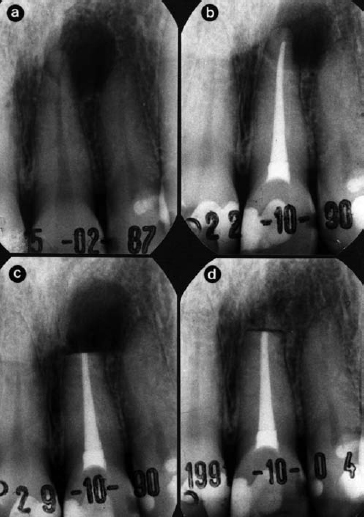
Fig 2. Radiographs of left maxillary central incisor followed over >4.5 year period. A radiolucent lesion was present (a), which was managed by endodontic treatment. The tooth was followed-up over 44 months with no signs of healing (b), so periapical surgery was performed (c) and 1 year later there is complete bone repair (d). Histopathological analysis of the surgical specimen revealed a cyst filled with cholesterol crystals. No bacteria were detected in the root apex or lesion. Reprinted with permission from Nair.142
Flora in root-filled canals
It is generally acknowledged that persistence of disease is most commonly due to difficulties that occur during initial endodontic treatment. Inadequate aseptic control, poor access cavity design, missed canals, inadequate instrumentation, and breakdown of temporary or permanent restorations are examples of procedural pitfalls that may result in persistence of endodontic disease (Fig 1).42
The reasons for disease persistence in well-treated root-filled teeth have been poorly characterized until a series of studies published during the 1990s.43-46 Using block biopsy material from non-healed periapical tissues including apices of the root-filled teeth, analysis by correlative light and electron microscopy showed that there were four factors that may have contributed to persistence of a periapical radiolucency after treatment (Fig 2). The factors were: (i) intraradicular infection;43 (ii) extraradicular infection by bacteria of the species Actinomyces israelii and Propionibacterium propionicum;47-49 (iii) foreign body reaction;44,50 (iv) cysts, especially those containing cholesterol crystals.45
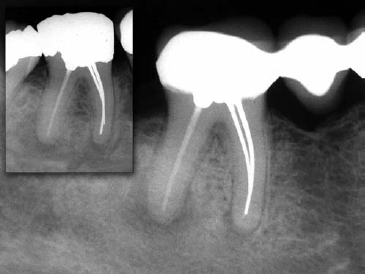
Fig 3. Apical periodontitis may continue for many years without symptoms. Main radiograph shows lower right molar which had received endodontic treatment 15 years ago. Inset shows same tooth 10 years prior to main radiograph. The unsatisfactory root canal treatment should have been re-done before the tooth was utilized as a bridge abutment.
Rarely, healing may occur by fibrous scar tissue instead of by bone,46 which may be misinterpreted as disease persistence on follow-up radiographs. Of all these factors, it is generally acknowledged that the major cause of post-treatment disease is the persistence of micro-organisms in the apical part of root-filled teeth.
Persistent endodontic disease, or apical periodontitis associated with a root-filled tooth, can continue for many years and may become apparent only when a tooth requires a new restoration or is detected on a routine radiograph (Fig 3). The fact that some microorganisms are capable of survival under harsh, nutrient-limited conditions of the root-filled canal for so long is remarkable. Yet, little information was known about the micro-organisms involved in persistent intra-canal infection after root filling until 1998, when two studies revealed that the microbial flora associated with persistent endodontic disease is quite unlike that found in other oral infections, or that of the untreated root canal.51,52
Microbiology of canals with persistent infection
Usually one or just a few species are recovered from canals of teeth with persistent disease. These are predominantly Gram-positive micro-organisms and there is an equal distribution of facultative and obligate anaerobes.51,52 This microbial flora is distinctly different from infections in untreated root canals, which typically consists of a polymicrobial mix with approximately equal proportions of Gram-positive and Gram-negative species, dominated by obligate anaerobes.
There is some diversity of species isolated from root-filled teeth with persistent periapical disease, but there is a consensus amongst most studies that there is a high prevalence of enterococci and streptococci.51-57 Other species found in higher proportions in individual studies are lactobacilli,51 Actinomyces species and peptostreptococci55 and Pseudoramibacter alactolyticus, Propionibacterium propionicum, Dialister pneumosintes, and Filifactor alocis57 and Candida albicans.51-53,55-57 Some bacteriological findings from studies of root-filled teeth with persistent disease are shown in Table 1.
There is a difference in the microbial flora between poorly treated and well treated teeth when the canals are sampled at re-treatment. In poorly root-filled teeth, the flora is similar to the polymicrobial infection seen in untreated root canals,52,58 which is not surprising when viewed in the context of the likely reasons for the unsatisfactory treatment — inadequate aseptic methods and poor coronal restoration — that together allow an influx of carbohydrates and possibly new bacteria from the oral cavity.
The prevalence of enterococci has been a conspicuous finding in all studies that have investigated the flora in root-filled teeth,51-58 with one exception,59 and implicates Enterococcus faecalis as an opportunistic pathogen in persistent apical periodontitis.
Ecological differences between untreated and root-filled root canals
The untreated infected root canal is an environment that provides micro-organisms with nutritional diversity in a shifting pattern over time. The available nutrients are mainly peptides and amino acids, which favour anaerobic proteolytic species.
Table 1. Bacteriological findings in root-filled teeth with persistent periapical lesions
| Study | Species per root canal with bacteria | Enterococcus sp.* | Streptococcus sp.* | Candida sp.* | Actinomyces sp.* |
| Möller53 | 1.6 | 29 | 16 | 3 | ND |
| Molander et al.51 | 1.7 | 47 | 20 | 4 | 3 |
| Sundqvist et al.52 | 1.3 | 38 | 25 | 8 | 13 |
| Hancock et al.55 | 1.7 | 32 | 21 | 3 | 27 |
| Peciuliene et al.56 | 1.6 | 64 | — | 18 | — |
| Cheung & Ho59 | 2.6 (1.8 ‡) | ND | 50 | 17 | ND |
| Pinheiro et al.58 | 2.1 (1.8 ‡) | 55 | 33 | 4 | 20 |
| Siqueira & Rôças57 † | 4.1 | 77 | 23 | 9 | 5 |
*Per cent prevalence, in canals with micro-organisms.
†Identification by PCR. All other studies by culture.
‡Excluding poorly filled root canals.
ND Not detected.
Whilst the microbial flora in an untreated infected root canal may experience feast, in the well-filled root canal there is predominantly famine. Most or all of the original necrotic pulp will have been eliminated leaving dry, barren conditions for surviving microbial cells. These microbes endure a static environment and starvation, but with some luck may encounter a serumlike fluid transudate from the periapical tissue. The species that persist are those that either have survived the antimicrobial treatment, or have entered during treatment and found it possible to establish where others cannot do so. Where the coronal seal is defective or missing, there is the possibility for new infection of the root canal space.
Properties of species associated with persistent endodontic disease
With the exception of Actinomyces, which is primarily involved in extraradicular infection, other species commonly associated with persistent intraradicular infection such as candida and enterococci can be viewed as opportunistic pathogens. A shared behaviour is that they leave their normal habitat in the oral cavity and establish in the root canal where they take advantage of the ecological changes and that their microbial competitors have been eliminated by treatment.
For microbes to maintain apical periodontitis and continue to cause disease, they must do more than just survive in the root-filled canal; they must also possess the pathogenic properties necessary to perpetuate inflammation external to the root canal system. In general, micro-organisms involved in persistent infections implement one of three strategies to evade the immune response — sequestration, cellular or humoral evasion.60 Sequestration involves a physical barrier between the microbe and the host. Cellular evasion means that micro-organisms avoid leukocyte-dependent antibacterial mechanisms. Humoral evasion means that those extracellular bacteria avoid the host’s antibodies and complement.
At least two of the three strategies are deployed by micro-organisms involved in persistent endodontic disease.61 A. israelii is an example of an endodontic pathogen that displays cellular evasion by avoiding phagocytosis by PMN leukocytes in vivo62-64 primarily through a mechanism of collective cohesion.63 E. faecalis and Candida species are representative of microbes that can remain sequestered within the root canal system.
Micro-organisms involved in persistent endodontic disease require a range of properties that allow them to enter and establish in the root canal, survive the antimicrobial treatment and induce or maintain apical periodontitis (Fig 4). That low numbers of cells survive endodontic treatment implies an ability of some species to withstand instrumentation and antimicrobial irrigation, however numerous reports confirm the bactericidal efficacy of sodium hypochlorite against species involved in persistent infection such as A. israelii,65 E. faecalis66-68 and Candida.69-71 Thus, these species may have the capacity to shelter from the main root canal in web-like areas, canal ramifications or dentinal tubules where some level of protection or buffering of the antimicrobial agent is possible.72,73 Although most root canal bacteria are sensitive to the high pH of calcium hydroxide,74 several species involved in persistent infection are known to have a capacity to resist a high pH.68,74-79
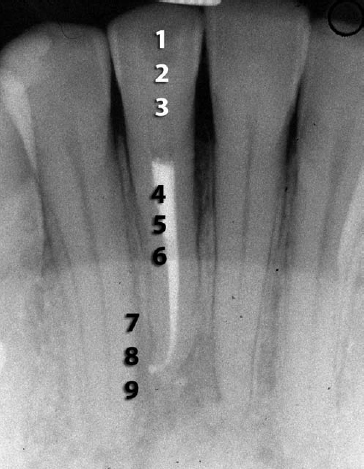
Fig 4. Challenges for microbes involved in persistent infection. There are three phases involved for micro-organisms to enter, persist and survive in the root-filled canal.
Entry and establishment. Microbes must (1) enter the root canal space, (2) compete against other members of the microbial flora and (3) acquire nutrition.
Survival of the antimicrobial treatment. Microbes must survive (4) cleaning, (5) root filling and then (6) endure starvation.
Maintaining apical periodontitis. Microbes must (7) have the capacity to utilize serum-like tissue fluid for nutrition, (8) survive the host defence and (9) be able to induce and maintain inflammation in periapical tissue.
How bacteria endure root filling is unknown, but studies that have sampled the root canal prior to root filling and then followed the treatment outcome of infected teeth have shown that some lesions heal,52,80-82 implying that the bacteria did not survive or were not able to inflame the periapical tissue. Whether or not bacteria survive root filling may depend on whether they are entombed, or blocked from acquiring nutrition. It is possible, even likely, that bacteria may undergo a period of starvation. The ability of E. faecalis to endure periods of starvation,83-86 is a trait that may be crucial for survival.
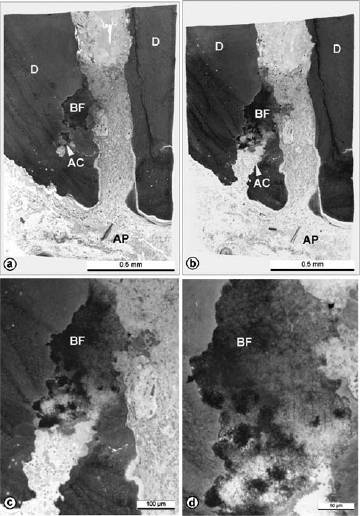
Fig 5. The location of microbes is critical for access to host-derived nutrients and to inflame periapical tissue. Light microscopic view of a surgically removed root apex with persistent apical periodontitis, showing proximity of a microbial mass in communication with periapical tissue. Consecutive sections (a, b) reveal the emerging widened profile of an accessory canal (AC) that is clogged with microbial biofilm (BF). The accessory canal and biofilm are magnified in (c) and (d) respectively. Reprinted with permission from Nair et al.43
Apical periodontitis is a dynamic process involving an interaction between host and living bacteria, and the microbes need to find substrates for growth. In a well-instrumented root canal where necrotic pulp tissue has been removed and there is no communication with exogenous nutrients from the oral cavity, nutrition is likely to come from a periapical fluid transudate, which is probably serum-like in nature. The capacity of some species to degrade serum and tissue molecules corresponds with an ability to avoid the host defence and induce an inflammatory response.87 An ability to utilize collagen within dentine may also be useful and there are indications that E. faecalis may have this property.88,89
Conditions for persistent infection
In a study that examined the influence of infection at the time of root filling on the outcome of treatment,80 68 per cent of teeth that were infected at root filling healed after the treatment. Similar results have also been reported in other studies.52,81,82 Whilst infection at the time of root canal filling will adversely affect the outcome of treatment, the mere presence of an endodontic pathogen is not in itself sufficient for disease persistence. Several parameters must be met for micro-organisms to maintain apical periodontitis following endodontic treatment.
Persistent endodontic treatment disease involves multiple microbial and location factors. Microorganisms must possess an ability to survive the antimicrobial treatment and require ‘persistence’ characteristics such as a capacity for starvation survival and an ability to utilize serum-like periapical transudate as a nutritional source. The location of microbes within the root canal system is crucial for access to nutrients. They must be situated near the apical (or an accessory) foramen and have an open communication for the free exchange of fluid, molecules and for organisms to inflame the periapical tissue (Fig 5). Together, these microbial characteristics and opportunities of location determine whether microorganisms that survive treatment are able to maintain apical periodontitis following such treatment.
Importance of asepsis
Since the primary goal of endodontic therapy is the elimination of bacteria from the root canal, an essential requirement during treatment is that it be undertaken in a sterile environment where further contaminating micro-organisms can be reliably excluded from the canal. The treatment of root canal infections is unique in the sense that it is possible to isolate the area from the rest of the oral cavity by use of rubber dam. Efficient methods are available for disinfection of the operative field, the tooth and rubber dam. The importance of using sterile instruments and an aseptic technique in a disinfected field cannot be overemphasized, since failure to do so may have a direct bearing on the outcome of treatment.
Every effort should be made to exclude and eliminate micro-organisms from the operative field and the root canal itself. Application of the rubber dam is mandatory for endodontic treatment. The rubber dam must properly isolate the tooth from the oral cavity to ensure an aseptic field.
Once the tooth is isolated from the oral cavity, the tooth surface and adjacent rubber dam should be cleaned with 30% hydrogen peroxide, taking care to ensure that the skin and eyes of the patient and staff are protected. The same area is then carefully disinfected by scrubbing the area with 5% tincture of iodine,53 sodium hypochlorite90 or 2% chlorhexidine in alcohol. These procedures are simple yet effective and greatly reduce the chances of contaminating the open root canal.
Antimicrobial effect of debridement
The control of bacteria within the root canal might appear to be straightforward since such a large proportion of the bacterial flora is sensitive to oxygen. However, the penetration of oxygen into the canal during treatment does not seem to have any significant effect on the bacteria. The reason for this is that many of the bacteria are protected in the irregularities and branches of the root canal system and in dentinal tubules. Only a few cells need to survive treatment so that when the canal is closed, the anaerobic milieu will be restored and the bacteria can re-multiply. The microbial flora within the root canal must be actively eliminated by a combination of physical debridement and antimicrobial chemical treatment.
Although the most important aspect of root canal instrumentation is undoubtedly the elimination of bacteria and the removal of remnants of pulp tissue and debris, the shaping of the root canal to accommodate the root filling material is also of importance. A great deal has been written about the preparation of the root canal to achieve a tapering form91 since this shape facilitates cleaning of the apical third,92 preserves the apical foramen from over-instrumentation and facilitates filling of the canal.
Preparation of the root canal consists of two main phases: debridement by manual and mechanical instrumentation, and chemical disinfection by irrigation and subsequent antibacterial dressing. The relative effectiveness of these measures has been studied in a series of investigations with advanced bacteriological techniques74,93-97 and the cleansing effect of the mechanical instrumentation has been studied by histology92,98,99 and by scanning electron microscope (SEM) analysis of the appearance of the root canal wall before and after instrumentation of the root canal. 72,100-102
Antimicrobial efficacy of manual instrumentation
Infected root canals can harbour between 102 to more than 109 bacterial cells.93,97 Manual instrumentation with 6–10ml of saline per canal can reduce the number of bacteria in infected root canals by 100 to 1000-fold.93 However, root canal preparation with hand files and saline irrigation is only moderately effective. Early studies in which no antiseptic irrigants were used reported that 20 to 30 per cent of the root canals that were infected at the beginning of treatment yielded negative cultures at the end of the first appointment.103-105 These studies are of limited value because they were performed with bacteriological techniques unsuited to the recovery of anaerobes.
Using advanced bacteriological techniques, it has been shown that the number of bacteria can be significantly reduced, but not to an extent that negative cultures can be reliably obtained at the end of the first appointment.93 This underlies the importance of supporting the mechanical cleaning of canals with antimicrobial irrigation.
SEM studies of canals that have been manually instrumented with saline irrigation show that loose debris can be eliminated from the upper and middle thirds of the root canal.101 Filing with endodontic instruments translocates and burnishes the superficial components (organic and inorganic) of the circumpulpal dentine and creates an amorphous smear layer on the canal walls.106-108 This layer is not affected by irrigation with saline.101
Table 2. Effect of stainless steel or NiTi instrumentation on bacterial elimination
| Instrumentation and irrigation regime | Canals rendered bacteria free % | |
| Sjögren et al.97 | St/steel, NaOCl | 50 |
| Sjögren et al.80 | St/steel, NaOCl | 60 |
| Shuping et al.109 | NiTi, NaOCl | 62 |
| McGurkin-Smith et al.110 | NiTi, EDTA + NaOCl | 53 |
| Waltimo et al.111 | St/steel, NaOCl | 76 |
Antimicrobial efficacy of rotary NiTi instrumentation
Engine-driven instruments make canal preparation faster and less tedious than hand instrumentation. The flexibility of nickel-titanium rotary instruments facilitates shaping of curved canals and enables the clinician to instrument canals to the desired tapered form with a high degree of consistency.
The antimicrobial effectiveness of instrumenting canals with rotary nickel-titanium instruments is in line with the results seen with manual instrumentation of root canals.80,97,109-111 Thus, instrumentation with either stainless steel or nickel-titanium rotary instruments in the presence of NaOCl renders canals free of bacteria in half to three-quarters of cases (Table 2). A recent in vivo study that applied correlative light and electron microscopic techniques to evaluate residual intracanal infection after instrumentation with stainless steel hand files in mesiobuccal canals and NiTi instruments in mesiolingual canals of the same lower molars showed that there was no difference in their respective ability to eliminate infection.99 That bacteria cannot be completely eliminated after thorough instrumentation and irrigation regardless of the technique (Fig 6) points to the need to follow instrumentation with an antibacterial dressing before obturation to better achieve the goal of bacteria-free root canals.
Does apical enlargement eliminate infection?
A number of studies have suggested that apical enlargement may reduce the microbial flora compared with instrumentation to smaller file sizes. These studies, some of which are listed in Table 3, have been performed under different conditions with divergent results. On the basis of selected findings, some authors have advocated that it should then be possible to complete endodontic treatment in one visit if cleaning to a large apical size completely eradicates bacteria.
With significant root canal enlargement, it is not surprising that it might result in a reduction in the bacterial flora, but it happens at the expense of tooth structure. Can this method of instrumentation totally and predictably eliminate bacteria, or is it simply a further reduction of the overall bacterial load? It should be appreciated that whilst bacterial reduction is undoubtedly desirable, the goal of endodontic treatment is bacterial eradication. Whenever bacterial cells survive, there remains a risk that they may multiply and reach numbers that have the capacity to maintain periapical inflammation.
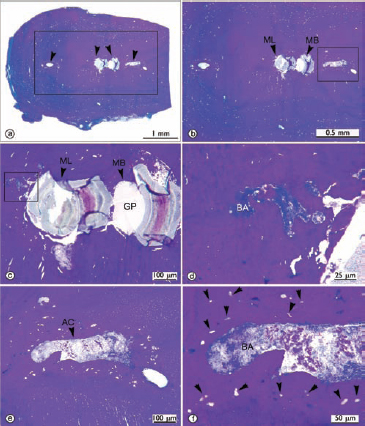
Fig 6. Microbes grow as biofilms in the root canal system and may be located in areas that are inaccessible to instrumentation such as isthmuses or accessory canals. Light microscopic view of transverse section through a surgically removed root apex of a mesial root of lower molar (a). The rectangular demarcated area in (a) is magnified in (b) and the mesiolingual (ML) and mesiobuccal (MB) canals, magnified in (c), communicate and are root-filled (GP). The rectangular demarcated area in (b) is magnified in (e). The main canals show recesses and diverticulations; those in the rectangular demarcated area in (c) are magnified in (d). One non-instrumented accessory canal (AC in (e)) is enlarged in (f). The larger accessory canal in (e, f) is clogged with bacteria (BA). Black arrowheads in (f) show cross-sectioned profiles of anastomoses of the root canal system. Reprinted with permission from Nair et al.99
It is known that after instrumentation there are very few remaining cells, which may number less than 102 and special procedures and precautions are required to recover the remaining bacterial cells. Only a few culture studies have adopted the measures necessary to recover these cells. As an example, the steps taken in one of these studies80 for recovery of small numbers of cells after instrumentation is shown in Table 4. In contrast, a study112 cited as showing that increased apical enlargement results in bacterial elimination claimed that 89 to 100 per cent of canals were rendered bacteria free when molars were instrumented to a size 60 and canines/premolars to a size 80, respectively. The bacteriological methods for sampling after instrumentation and culture in that study are shown in Table 4.
Table 3. Effect of larger apical sizes on apparent bacterial reduction
| Type | No. of bacteria | |
| Yared & Bou Dagher143 | In vivo | Reduction |
| Siqueira et al.144 | In vitro | Reduction |
| Shuping et al.109 | In vivo | Reduction |
| Rollison et al.145 | In vitro | Reduction |
| Coldero et al.146 | In vitro | No difference |
| Card et al.112 | In vivo | Elimination |
When a sensitive technique is used80 the chances are optimized for bacterial recovery. For example, where three samples are taken and no growth is seen with one sample the others can be used as backup and check of the first sample. An ‘enrichment’ medium, fluid thioglycolate medium (FTM), was used so that if colonies did not appear on the plates, new plates were inoculated from the other PYG broth and the FTM. If none are positive for bacteria, one can be more assured of the veracity of a negative result. Even the application of paper points must be done with utmost care to maximize recovery of fluid from the root canal. Figure 7 shows two paper points from the same canal. One is negative and the other is positive for bacteria, because the left one is thinner and reached further apically in the canal to capture bacteria. The light microscopic view of a sectioned tooth root apex shown in Fig 5 illustrates where bacteria may be located and how they can escape recovery if a paper point does not reach the full canal length.
Dilution of a sample is another critical step in bacterial cultivation. The mean number of cells in an untreated case may be as high as 106 to 109cfu/ml and in order to quantify cells and distinguish species from one another, serial dilution is necessary to obtain about 50–200cfu per agar plate. However, when small numbers of cells and species are anticipated in a sample, such as after antimicrobial instrumentation or filling, the method of dilution is markedly different. Under these conditions, minimal dilution and large aliquots of the sample inoculated onto the plate are more likely to recover a few cells than a small aliquot and an automated spiral plate dilution device (Table 4).
Other factors may also influence recovery of cells. After instrumentation, the surviving bacteria are in a fragile state and are vulnerable to handling and oxygen exposure. Thus, handling the sample in an anaerobic box and growth on media for at least 10d enhances the chances for cell survival. Identification of recovered species helps ensure sample accuracy and protects against contamination. Thus, recovery of species not normally identified in the root canal, e.g., Staphylococcus epidermidis, would imply a contaminant and this provides a check of sample integrity. Similarly, tracing a species from sample to sample through a single clinical case helps ensure that a species isolated after treatment was present in the canal from the beginning of treatment. If a species is identified in a post-treatment sample that has not been isolated in pretreatment samples, it implies contamination. Simple bacterial counts without species identification cannot provide the same level of information.
Table 4. Examples of differences in sampling method that influence microbe recovery
| Sjögren et al.80 | Card et al.112 |
| 3 samples: 2 in PYG broth, 1 in
enrichment FTM Large aliquot on plate, no dilution Anaerobic box, >10 d. If no growth, used backups Species identification |
1 sample: Liquid DTF Small volume on plate, spiral plate dilution Anaerobic gas jar, incubated 5–7 d Simple counts. No species identification |
Without a deeper insight of bacteriological procedures, these differences may appear slight yet the consequences of a less sensitive method for cultivation and culturing are that it is unlikely that small numbers of cells will be identified in a sample. This is likely to account for the perceived differences in recovery of bacteria from cleaned root canals and limits the conclusions that can be drawn from such material. That complete eradication of infection cannot be achieved by apical enlargement is confirmed by immunohistological113 and microscopic analysis of canals instrumented in lower molars,99 which revealed that microbes are often located as biofilms in inaccessible areas of the canal system such as isthmuses or accessory canals (Fig 6). Thus, on the basis of currently available information there is insufficient scientific support for the idea that it is possible to eliminate infection by apical enlargement of the canal space.
Clinical implications of apical enlargement
Even though the use of rotary NiTi instrumentation allows curved roots to be widened to sizes 45 to 80, there is a question about the effect of such enlargement on tooth structure. More than 25 years ago, cleaning to large apical sizes was advocated114 and was fraught with procedural clinical problems, mainly associated with iatrogenic damage to the fine structure of the apical third of the root. The difference between the stainless steel instruments of that time and NiTi instruments of today is that the increased flexibility of NiTi instruments reduces the chance of deviation from the original canal anatomy during instrumentation. However, an open question is: compared with conservative techniques of apical preparation, does apical enlargement provide better clinical results and with a suitable margin of safety?

Fig 7. Two paper points from the same canal. The right one is negative for bacteria, whereas the left paper point is thinner and has reached further apically in the canal to recover bacteria.
Observation of the results of those practising techniques of apical enlargement has shown that, apart from in the hands of the most highly skilled clinicians, there are many procedural risks associated with apical enlargement. Some of the potential procedural problems associated with enlarged apical instrumentation are summarised in Table 5.
As a direct consequence of apical enlargement, the tooth structure surrounding the root canal space is thinned and the risk to the integrity of the root structure is greatest where there is the smallest amount of original tooth structure. Consequently, the risk of apical perforation is progressively higher with increasing instrument size and may result in root weakness and splitting of the delicate apical root structure.
A lower margin of safety applies with apical enlargement since a minor miscalculation in measurement may result in a significantly more damaged apex. An over-instrumentation error with a size 60 or 80 file leaves a much larger apical wound than a size 25 file — the wound surface area is 6–10 times greater with a size 60 or 80 instrument than a size 25 file, respectively. Thus, with apical enlargement the operator skill becomes a much more critical factor for the outcome of clinical treatment and there is a noticeably lower margin of safety.
Apart from the risks to the integrity of tooth structure, apical enlargement also has the potential to adversely impact on subsequent endodontic procedures. Once the canal has been instrumented to a larger size (>#45), the filling material is more difficult to control during obturation. If, in addition, the apex has been opened there is a higher risk of overfilling, which has been shown to be associated with a reduced success rate.114-117 Lastly, if an apically enlarged root filling fails, it may be much more difficult or impossible to successfully retreat the tooth. This is because the more dentine removed during initial treatment, the less dentine is available for preparation at re-treatment. Further instrumentation of an apically enlarged canal significantly increases both the risk of procedural errors (perforation, zipping, etc.) and heightens the risk of excessive apical root weakness or splitting.
Table 5. Potential pitfalls in apical enlargement (see text for details)
| Risk | Reason |
| Apical perforation Root weakness & apical splitting Low margin of error Challenging to root fill Difficult to retreat |
Higher risk of perforation due to larger
apical size Thinned root canal walls and reduced apical tooth structure Minor errors in measurement, may result in a damaged apex Filling material is more difficult to control during obturation Thin root walls may preclude or hamper retreatment |
Based on current knowledge, the answer to the question ‘does increased apical enlargement predictably eliminate bacteria?’ is no. To the question ‘does apical enlargement provide better clinical results and with a suitable margin of safety?’, the answer is no. The results achievable with a suitable antimicrobial dressing are more predictable and do so in a conservative way. Taken together, the thin evidence for apical enlargement as a means of bacterial eradication, and the significantly increased risk of procedural errors, the disadvantages and risks of apical enlargement far outweigh the perceived benefits.
Antimicrobial effect of chemical agents
The antibacterial effect of mechanical preparation with saline as an irrigant has been shown to be inefficient in the elimination of bacteria from the root canal.93,118 This implies that mechanical instrumentation of the canal must be supplemented by antibacterial irrigants and dressings for efficient elimination of micro-organisms from the root canal. There are many other benefits to be gained by the use of chemical agents during the preparation of the root canal. The agents used for chemical disinfection can be separated into two types — those used for irrigation during canal preparation and those used as an intracanal dressing between appointments.
Irrigation
Irrigation of the root canal is an essential component of root canal preparation. The main benefits of using irrigants during the cleaning of the canal include wetting of the canal walls and removal of debris by flushing, destruction of micro-organisms, dissolution of organic matter, removal of smear layer and softening of dentine and cleaning in areas that are inaccessible to mechanical cleansing methods.
When applied to infected tissue, an irrigant should ideally destroy micro-organisms and their toxins without damaging normal tissues. Sodium hypochlorite solution (NaOCl) was identified early last century as a promising microbicide that did not cause tissue damage or interfere with wound healing.119
A clearly superior antibacterial effect has been demonstrated when NaOCl solution is used as an irrigant. Although the concentration of NaOCl solution seems to have little apparent effect on antimicrobial activity in the root canal system,95,120 the efficacy of weak solutions decreases rapidly and consequently irrigation should be frequent and copious. In addition to its powerful antimicrobial activity, NaOCl solution has a strong capacity for dissolution of organic matter.101,102,121-123 The tissue dissolving ability of NaOCl solution is influenced by the amount of organic matter, the fluid flow around this matter and the surface area available for interaction.124
The chelating agent ethylenediamine tetra-acetic acid (EDTA) is commonly used as an irrigant in conjunction with NaOCl solution, because EDTA is highly effective in removal of the smear layer and opening dentinal tubules,101 which potentiates the reach of antimicrobial irrigation and dressing.
There are many regions of the root canal system that are simply inaccessible to mechanical instrumentation and all of these areas have the potential to harbour micro-organisms and necrotic pulp tissue. These areas include accessory canals, fins and webs that branch from the main canal or canals. Areas that are inaccessible to mechanical instrumentation can only be cleaned by antimicrobial irrigants that are able to permeate into these recesses. Any further antibacterial effect will only occur with the support of an intracanal dressing.
Dressings
Mechanical cleaning, irrigation and dressing with antibacterial medicaments achieve a reduction of the number of living bacteria in infected root canals. Evaluation of the relative efficacy of these measures in eliminating the bacteria has shown that mechanical cleansing supported by irrigation significantly reduces the number of bacteria in the root canal, but that approximately 25 to 50 per cent of canals treated in this way still contain bacteria at the end of the appointment.80,97,109-111 The number of persisting bacterial cells is usually low, but these remaining bacteria can recover and rapidly increase in number between treatment visits if no antibacterial dressing is present in the root canal. The growth of bacteria between appointments can ultimately lead to the re-establishment of the number of bacteria that were initially present in the root canal before treatment.
The principal goal of dressing the root canal between appointments is to ensure a safe, antibacterial action with a long-lasting effect. If the active agent in the medicament is rapidly lost then the duration of its antibacterial activity is likely to be short, and thus ineffective. The antibacterial effect of dressing root canals with camphorated paramonochlorophenol and camphorated phenol has been assessed in vivo and been shown to be of limited efficacy.74
The clinical effectiveness of calcium hydroxide in infected canals has been tested in a number of in vivo studies and been shown to be an efficient antibacterial treatment eliminating micro-organisms in previously untreated cases from 75 per cent125-127 to more than 90 per cent of dressed canals.74,97,109,128 Application of an interappointment calcium hydroxide dressing prior to obturation has been shown to yield improved healing responses over non-calcium hydroxide treated teeth in human81 and animal teeth.82,129-132 Treatment with calcium hydroxide has also been shown to dissolve necrotic tissue and enhance the tissue dissolving effect of NaOCl solution.122,133-135
It is critical that the calcium hydroxide dressing be placed carefully in the instrumented canal as a thick, moist paste fully filling the entire canal136 and that it be left for sufficient time to achieve the desired antimicrobial effect. The paste consistency helps prevent influx of the periapical fluid, which is an important nutrient source for any remaining bacterial cells. The hydroxyl ions that are responsible for the strong antibacterial effect are rapidly potent when in intimate contact with target micro-organisms in vitro,65,74,137 but need time under in vivo conditions to diffuse into the adjacent dentine. The reason for the slow diffusion of hydroxyl ions into dentine is the powerful buffering capacity of dentine, which creates a concentration gradient across the root wall.136,138-141 Dressing the canal for one week has been shown to be an efficient method in the clinical setting.97
After canal preparation and final irrigation, residual fluid should be aspirated leaving the canal walls moist since the antimicrobial effectiveness of calcium hydroxide depends on an aqueous environment. Calcium hydroxide is easily applied as a paste into the canal with a spiral paste filler. Any residual calcium hydroxide on the walls of the access cavity should be carefully removed before the temporary filling is placed. A well placed temporary filling of >4mm depth is essential, for without it the many antibacterial steps preceding its placement are rapidly undone.
CONCLUSION
Infection of the root canal is not a random event. The type and mix of the microbial flora develop in response to the surrounding environment. Factors that influence whether species die or survive are the particular ecological niche, nutrition, anaerobiosis, pH and competition or cooperation with other microorganisms. Whether it is a necrotic pulp or root-filled space, the environment selects for micro-organisms that possess traits suited to establishing and sustaining the disease process.
Reduction and elimination of micro-organisms from the infected root canal provides the optimal chance of treatment success. The goal of achieving a clean, microbe-free canal can best be realised by working in a sterile way using instrumentation with antibacterial irrigation, which is reinforced by an intracanal dressing with calcium hydroxide. Alternating regimes of sodium hypochlorite solution and EDTA during canal preparation followed by dressing with calcium hydroxide for a minimum of seven days are a powerful combination for elimination of bacteria from the root canal. Provided sufficient time is available for canal preparation at the first visit and filling of canals at the second, endodontic treatment can be completed in just two visits with the assurance that there is a high chance of eliminating bacteria from the root canal system.
REFERENCES
1. Marsh PD. Are dental diseases examples of ecological catastrophes? Microbiology 2003;149:279-294.
2. Mims C, Dimmock N, Nash A, Stephen J. Mims’ pathogenesis of infectious disease. 4th edn. London: Academic Press, 1995.
3. Moore WEC, Moore LVH. The bacteria of periodontal diseases. Perio 2000 1994;5:66-77.
4. Paster BJ, Boches SK, Galvin JL, et al. Bacterial diversity in human subgingival plaque. J Bacteriol 2001;183:3770-3783.
5. Kantz WE, Henry CA. Isolation and classification of anaerobic bacteria from intact pulp chambers of non-vital teeth in man. Archs Oral Biol 1974;19:91-96.
6. Wittgow WC, Jr., Sabiston CB, Jr. Microorganisms from pulpal chambers of intact teeth with necrotic pulps. J Endod 1975;1:168-171.
7. Sundqvist G. Bacteriological studies of necrotic dental pulps. Umeå University Odontological Dissertations No. 7. Umeå: Umeå University, Sweden, 1976.
8. Sundqvist G. Taxonomy, ecology, and pathogenicity of the root canal flora. Oral Surg Oral Med Oral Pathol 1994;78:522-530.
9. Fabricius L, Dahlén G, Öhman AE, Möller ÅJR. Predominant indigenous oral bacteria isolated from infected root canals after varied times of closure. Scand J Dent Res 1982;90:134-144.
10. Loesche WJ. Importance of nutrition in gingival crevice microbial ecology. Periodontics 1968;6:245-249.
11. Carlsson J, Frölander F, Sundqvist G. Oxygen tolerance of anaerobic bacteria isolated from necrotic dental pulps. Acta Odont Scand 1977;35:139-145.
12. Loesche WJ, Gusberti F, Mettraux G, Higgins T, Syed S. Relationship between oxygen tension and subgingival bacterial flora in untreated human periodontal pockets. Infect Immun 1983;42:659-667.
13. Lev M, Keudell KC, Milford AF. Succinate as a growth factor for Bacteroides melaninogenicus. J Bacteriol 1971;108:175-178.
14. Grenier D, Mayrand D. Nutritional relationships between oral bacteria. Infect Immun 1986;53:616-620.
15. Marsh PD. Host defenses and microbial homeostasis: role of microbial interactions. J Dent Res 1989;68:1567-1575.
16. Ohta H, Kato K, Fukui K, Gottschal JC. Microbial interactions and the development of periodontal disease. J Periodontal Res 1991;26:255-257.
17. Jansen H-J, van der Hoeven JS. Protein degradation by Prevotella intermedia and Actinomyces meyeri supports the growth of non-protein-cleaving oral bacteria in serum. J Clin Periodontol 1997;24:346-353.
18. Gibbons RJ, Engle LP. Vitamin K compounds in bacteria that are obligate anaerobes. Science 1964;146:1307-1309.
19. Carlsson J. Microbiology of plaque associated periodontal disease. In: Lindhe J, ed. Textbook of clinical periodontology. Copenhagen: Munksgaard, 1990:129-152.
20. Sundqvist G. Associations between microbial species in dental root canal infections. Oral Microbiol Immunol 1992;7:257-262.
21. Sundqvist G. Ecology of the root canal flora. J Endod 1992;18:427-430.
22. Gomes BP, Drucker DB, Lilley JD. Positive and negative associations between bacterial species in dental root canals. Microbios 1994;80:231-243.
23. Lana MA, Ribeiro-Sobrinho AP, Stehling R, et al. Microorganisms isolated from root canals presenting necrotic pulp and their drug susceptibility in vitro. Oral Microbiol Immunol 2001;16:100-105.
24. Peters LB, Wesselink PR, van Winkelhoff AJ. Combinations of bacterial species in endodontic infections. Int Endod J 2002;35:698-702.
25. ter Steeg PF, van der Hoeven JS. Development of periodontal microflora on human serum. Microb Ecol Health Dis 1989;2:1-10.
26. Nair PNR. Light and electron microscopic studies of root canal flora and periapical lesions. J Endod 1987;13:29-39.
27. Costerton JW, Stewart PS. Biofilms and device-related infections. In: Nataro JP, Blaser MJ, Cunningham-Rundles S, eds. Persistent bacterial infections. Washington, DC: ASM Press, 2000:423-439.
28. Costerton W, Veeh R, Shirtliff M, Pasmore M, Post C, Ehrlich G. The application of biofilm science to the study and control of chronic bacterial infections. J Clin Invest 2003;112:1466-1477.
29. Abdullah M, Ng YL, Gulabivala K, Moles DR, Spratt DA. Susceptibilties of two Enterococcus faecalis phenotypes to root canal medications. J Endod 2005;31:30-36.
30. Woese CR. Bacterial evolution. Microbiol Rev 1987;51:221-271.
31. Jung IY, Choi B, Kum KY, et al. Identification of oral spirochetes at the species level and their association with other bacteria in endodontic infections. Oral Surg Oral Med Oral Pathol Oral Radiol Endod 2001;92:329-334.
32. Siqueira JF, Jr., Rôças IN, Favieri A, Oliveira JC, Santos KR. Polymerase chain reaction detection of Treponema denticola in endodontic infections within root canals. Int Endod J 2001;34:280-284.
33. Baumgartner JC, Khemaleelakul SU, Xia T. Identification of spirochetes (treponemes) in endodontic infections. J Endod 2003;29:794-797.
34. Rôças IN, Siqueira JF, Jr., Andrade AF, Uzeda M. Oral treponemes in primary root canal infections as detected by nested PCR. Int Endod J 2003;36:20-26.
35. Siqueira JF, Jr., Rôças IN. PCR-based identification of Treponema maltophilum, T amylovorum, T medium, and T lecithinolyticum in primary root canal infections. Arch Oral Biol 2003;48:495-502.
36. Conrads G, Gharbia SE, Gulabivala K, Lampert F, Shah HN. The use of a 16S rDNA directed PCR for the detection of endodontopathogenic bacteria. J Endod 1997;23:433-438.
37. Gonçalves RB, Mouton C. Molecular detection of Bacteroides forsythus in infected root canals. J Endod 1999;25:336-340.
38. Rôças IN, Siqueira JF, Jr., Santos KR, Coelho AM. “Red complex” (Bacteroides forsythus, Porphyromonas gingivalis, and Treponema denticola) in endodontic infections: a molecular approach. Oral Surg Oral Med Oral Pathol Oral Radiol Endod 2001;91:468-471.
39. Fouad AF, Barry J, Caimano M, et al. PCR-based identification of bacteria associated with endodontic infections. J Clin Microbiol 2002;40:3223-3231.
40. Siqueira JF, Jr., Rôças IN. Bacteroides forsythus in primary endodontic infections as detected by nested PCR. J Endod 2003;29:390-393.
41. Sundqvist G, Figdor D. Life as an endodontic pathogen. Ecological differences between the untreated and root-filled root canals. Endod Topics 2003;6:3-28.
42. Sundqvist G, Figdor D. Endodontic treatment of apical periodontitis. In: Ørstavik D, Pitt Ford TR, eds. Essential Endodontology Prevention and treatment of apical periodontitis. London: Blackwell Science, 1998:242-277.
43. Nair PNR, Sjögren U, Krey G, Kahnberg K-E, Sundqvist G. Intraradicular bacteria and fungi in root-filled, asymptomatic human teeth with therapy-resistant periapical lesions: a long-term light and electron microscopic follow-up study. J Endod 1990;16:580-588.
44. Nair PNR, Sjögren U, Krey G, Sundqvist G. Therapy-resistant foreign body giant cell granuloma at the periapex of a root-filled human tooth. J Endod 1990;16:589-595.
45. Nair PNR, Sjögren U, Schumacher E, Sundqvist G. Radicular cyst affecting a root-filled human tooth: a long-term post-treatment follow-up. Int Endod J 1993;26:225-233.
46. Nair PNR, Sjögren U, Figdor D, Sundqvist G. Persistent periapical radiolucencies of root-filled human teeth, failed endodontic treatments, and periapical scars. Oral Surg Oral Med Oral Pathol Oral Radiol Endod 1999;87:617-627.
47. Sundqvist G, Reuterving C-O. Isolation of Actinomyces israelii from periapical lesion. J Endod 1980;6:602-606.
48. Nair PNR, Schroeder HE. Periapical actinomycosis. J Endod 1984;10:567-570.
49. Sjögren U, Happonen RP, Kahnberg KE, Sundqvist G. Survival of Arachnia propionica in periapical tissue. Int Endod J 1988;21:277-282.
50. Koppang HS, Koppang R, Solheim T, Aarnes H, Stolen SO. Cellulose fibers from endodontic paper points as an etiological factor in postendodontic periapical granulomas and cysts. J Endod 1989;15:369-372.
51. Molander A, Reit C, Dahlén G, Kvist T. Microbiological status of root-filled teeth with apical periodontitis. Int Endod J 1998;31:1-7.
52. Sundqvist G, Figdor D, Persson S, Sjögren U. Microbiologic analysis of teeth with failed endodontic treatment and the outcome of conservative re-treatment. Oral Surg Oral Med Oral Pathol Oral Radiol Endod 1998;85:86-93.
53. Möller ÅJR. Microbiological examination of root canals and periapical tissues of human teeth. Methodological studies. Odontol Tidsk 1966;74:Suppl: 1-380.
54. Peciuliene V, Balciuniene I, Eriksen HM, Haapasalo M. Isolation of Enterococcus faecalis in previously root-filled canals in a Lithuanian population. J Endod 2000;26:593-595.
55. Hancock HH, Sigurdsson A, Trope M, Moiseiwitsch J. Bacteria isolated after unsuccessful endodontic treatment in a North American population. Oral Surg Oral Med Oral Pathol Oral Radiol Endod 2001;91:579-586.
56. Peciuliene V, Reynaud AH, Balciuniene I, Haapasalo M. Isolation of yeasts and enteric bacteria in root-filled teeth with chronic apical periodontitis. Int Endod J 2001;34:429-434.
57. Siqueira JF, Jr., Rôças IN. Polymerase chain reaction-based analysis of microorganisms associated with failed endodontic treatment. Oral Surg Oral Med Oral Pathol Oral Radiol Endod 2004;97:85-94.
58. Pinheiro ET, Gomes BP, Ferraz CC, Sousa EL, Teixeira FB, Souza-Filho FJ. Microorganisms from canals of root-filled teeth with periapical lesions. Int Endod J 2003;36:1-11.
59. Cheung GS, Ho MW. Microbial flora of root canal-treated teeth associated with asymptomatic periapical radiolucent lesions. Oral Microbiol Immunol 2001;16:332-337.
60. Nataro JP, Blaser MJ, Cunningham-Rundles S. Persistent bacterial infections: commensalism gone awry or adaptive niche? In: Nataro JP, Blaser MJ, Cunningham-Rundles S, eds. Persistent bacterial infections. Washington, DC: ASM Press, 2000: 3-10.
61. Figdor D. Microbial aetiology of endodontic treatment failure and pathogenic properties of selected species. Umeå University Odontological Dissertations No. 79. Umeå: Umeå University, Sweden, 2002. PhD thesis.
62. Brown JR, von Lichtenberg F. Experimental actinomycosis in mice. Arch Path 1970;90:391-402.
63. Figdor D, Sjögren U, Sörlin S, Sundqvist G, Nair PNR. Pathogenicity of Actinomyces israelii and Arachnia propionica: experimental infection in guinea pigs and phagocytosis and intra-cellular killing by human polymorphonuclear leukocytes in vitro. Oral Microbiol Immunol 1992;7:129-136.
64. Sumita M, Hoshino E, Iwaku M. Experimental actinomycosis in mice induced by alginate gel particles containing Actinomyces israelii. Endod Dent Traumatol 1998;14:137-143.
65. Barnard D, Davies J, Figdor D. Susceptibility of Actinomyces israelii to antibiotics, sodium hypochlorite and calcium hydroxide. Int Endod J 1996;29:320-326.
66. Siqueira Jr JF, Machado AG, Silveira RM, Lopes HP, De Uzeda M. Evaluation of the effectiveness of sodium hypochlorite used with three irrigation methods in the elimination of Enterococcus faecalis from the root canal, in vitro. Int Endod J 1997;30:279-282.
67. Spratt DA, Pratten J, Wilson M, Gulabivala K. An in vitro evaluation of the antimicrobial efficacy of irrigants on biofilms of root canal isolates. Int Endod J 2001;34:300-307.
68. Evans M, Davies JK, Sundqvist G, Figdor D. Mechanisms involved in the resistance of Enterococcus faecalis to calcium hydroxide. Int Endod J 2002;35:221-228.
69. D’Arcangelo C, Varvara G, De Fazio P. An evaluation of the action of different root canal irrigants on facultative aerobicanaerobic, obligate anaerobic, and microaerophilic bacteria. J Endod 1999;25:351-353.
70. Waltimo TM, Orstavik D, Siren EK, Haapasalo MP. In vitro susceptibility of Candida albicans to four disinfectants and their combinations. Int Endod J 1999;32:421-429.
71. Sen BH, Akdeniz BG, Denizci AA. The effect of ethylenediaminetetraacetic acid on Candida albicans. Oral Surg Oral Med Oral Pathol Oral Radiol Endod 2000;90:651-655.
72. Huque J, Kota K, Yamaga M, Iwaku M, Hoshino E. Bacterial eradication from root dentine by ultrasonic irrigation with sodium hypochlorite. Int Endod J 1998;31:242-250.
73. Haapasalo HK, Sirén EK, Waltimo TM, Ørstavik D, Haapasalo MP. Inactivation of local root canal medicaments by dentine: an in vitro study. Int Endod J 2000;33:126-131.
74. Byström A, Claesson R, Sundqvist G. The antibacterial effect of camphorated paramonochlorophenol, camphorated phenol and calcium hydroxide in the treatment of infected root canals. Endod Dent Traumatol 1985;1:170-175.
75. Haapasalo M, Ørstavik D. In vitro infection and disinfection of dentinal tubules. J Dent Res 1987;66:1375-1379.
76. Ørstavik D, Haapasalo M. Disinfection by endodontic irrigants and dressings of experimentally infected dentinal tubules. Endod Dent Traumatol 1990;6:142-149.
77. Safavi KE, Spångberg LSW, Langeland K. Root canal dentinal tubule disinfection. J Endod 1990;16:207-210.
78. Waltimo TM, Siren EK, Orstavik D, Haapasalo MP. Susceptibility of oral Candida species to calcium hydroxide in vitro. Int Endod J 1999;32:94-98.
79. Kalfas S, Figdor D, Sundqvist G. A new bacterial species associated with failed endodontic treatment: identification and description of Actinomyces radicidentis. Oral Surg Oral Med Oral Pathol Oral Radiol Endod 2001;92:208-214.
80. Sjögren U, Figdor D, Persson S, Sundqvist G. Influence of infection at the time of root filling on the outcome of endodontic treatment of teeth with apical periodontitis. Int Endod J 1997;30:297-306. Erratum in: Int Endod J 1998;31:148.
81. Trope M, Delano EO, Ørstavik D. Endodontic treatment of teeth with apical periodontitis: single vs. multivisit treatment. J Endod 1999;25:345-350.
82. Katebzadeh N, Sigurdsson A, Trope M. Radiographic evaluation of periapical healing after obturation of infected root canals: an in vivo study. Int Endod J 2000;33:60-66.
83. Giard JC, Hartke A, Flahaut S, Benachour A, Boutibonnes P, Auffray Y. Starvation-induced multiresistance in Enterococcus faecalis JH2-2. Curr Microbiol 1996;32:264-271.
84. Wendt C, Wiesenthal B, Dietz E, Ruden H. Survival of vancomycin-resistant and vancomycin-susceptible enterococci on dry surfaces. J Clin Microbiol 1998;36:3734-3736.
85. Figdor D, Davies JK, Sundqvist G. Starvation survival, growth and recovery of Enterococcus faecalis in human serum. Oral Microbiol Immunol 2003;18:234-239.
86. Sedgley CM, Lennan SL, Appelbe OK. Survival of Enterococcus faecalis in root canals ex vivo. Int Endod J 2005;38:735-742.
87. Jansen H-J. The periodontal microflora as a protein-dependent anaerobic degradation system. Nijmegen: University of Nijmegen, The Netherlands, 1996.
88. Love RM. Enterococcus faecalis—a mechanism for its role in endodontic failure. Int Endo J 2001;34:399-405.
89. Hubble TS, Hatton JF, Nallapareddy SR, Murray BE, Gillespie MJ. Influence of Enterococcus faecalis proteases and the collagen-binding protein, Ace, on adhesion to dentin. Oral Microbiol Immunol 2003;18:121-126.
90. Ng YL, Spratt D, Sriskantharajah S, Gulabivala K. Evaluation of protocols for field decontamination before bacterial sampling of root canals for contemporary microbiology techniques. J Endod 2003;29:317-320.
91. Schilder H. Filling root canals in three dimensions. Dent Clin North Am 1967;Nov:723-744.
92. Walton RE. Histologic evaluation of different methods of enlarging the pulp canal space. J Endod 1976;2:304-311.
93. Byström A, Sundqvist G. Bacteriologic evaluation of the efficacy of mechanical root canal instrumentation in endodontic therapy. Scand J Dent Res 1981;89:321-328.
94. Byström A, Sundqvist G. Bacteriologic evaluation of the effect of 0.5 percent sodium hypochlorite in endodontic therapy. Oral Surg Oral Med Oral Pathol 1983;55:307-312.
95. Byström A, Sundqvist G. The antibacterial action of sodium hypochlorite and EDTA in 60 cases of endodontic therapy. Int Endod J 1985;18:35-40.
96. Sjögren U, Sundqvist G. Bacteriologic evaluation of ultrasonic root canal instrumentation. Oral Surg Oral Med Oral Pathol 1987;63:366-370.
97. Sjögren U, Figdor D, Spångberg L, Sundqvist G. The antimicrobial effect of calcium hydroxide as a short-term intracanal dressing. Int Endod J 1991;24:119-125.
98. Svec TA, Harrison JW. Chemomechanical removal of pulpal and dentinal debris with sodium hypochlorite and hydrogen peroxide vs normal saline solution. J Endod 1977;3:49-53.
99. Nair PNR, Henry S, Cano V, Vera J. Microbial status of apical root canal system of human mandibular first molars with primary apical periodontitis after “one-visit” endodontic treatment. Oral Surg Oral Med Oral Pathol Oral Radiol Endod 2005;99:231-252.
100. Baker NA, Eleazer PD, Averbach RE, Seltzer S. Scanning electron microscopic study of the efficacy of various irrigating solutions. J Endod 1975;1:127-135.
101. Baumgartner JC, Mader CL. A scanning electron microscopic evaluation of four root canal irrigation regimens. J Endod 1987;13:147-157.
102. Baumgartner JC, Cuenin PR. Efficacy of several concentrations of sodium hypochlorite for root canal irrigation. J Endod 1992;18:605-612.
103. Ingle JI, Zeldow BJ. An evaluation of mechanical instrumentation and the negative culture in endodontic therapy. J Am Dent Assoc 1958;57:471-476.
104. Grahnén H, Krasse B. The effect of instrumentation and flushing of nonvital teeth in endodontic therapy. A clinical and bacteriological study. Odont Revy 1963;14:167-177.
105. Stewart GG, Kapsimalas P, Rappaport H. EDTA and urea peroxide for root canal preparation. J Am Dent Assoc 1969;78:335-338.
106. McComb D, Smith DC. A preliminary scanning electron microscopic study of root canals after endodontic procedures. J Endod 1975;1:238-242.
107. Moodnik RM, Dorn SO, Feldman MJ, Levey M, Borden BG. Efficacy of biomechanical instrumentation: a scanning electron microscopic study. J Endod 1976;2:261-266.
108. Lester KS, Boyde A. Scanning electron microscopy of instrumented, irrigated and filled root canals. Br Dent J 1977;143:359-367.
109. Shuping GB, Ørstavik D, Sigurdsson A, Trope M. Reduction of intracanal bacteria using nickel-titanium rotary instrumentation and various medications. J Endod 2000;26:751-755.
110. McGurkin-Smith R, Trope M, Caplan D, Sigurdsson A. Reduction of intracanal bacteria using GT rotary instrumentation, 5.25% NaOCl, EDTA, and Ca(OH)2. J Endod 2005;31:359-363.
111. Waltimo T, Trope M, Haapasalo M, Ørstavik D. Clinical efficacy of treatment procedures in endodontic infection control and one year follow-up of periapical healing. J Endod 2005;31:863-866.
112. Card SJ, Sigurdsson A, Ørstavik D, Trope M. The effectiveness of increased apical enlargement in reducing intracanal bacteria. J Endod 2002;28:779-783.
113. Matsuo T, Shirakami T, Ozaki K, Nakanishi T, Yumoto H, Ebisu S. An immunohistological study of the localization of bacteria invading root pulpal walls of teeth with periapical lesions. J Endod 2003;29:194-200.
114. Kerekes K, Tronstad L. Long-term results of endodontic treatment performed with a standardized technique. J Endod 1979;5:83-90.
115. Strindberg LZ. The dependence of the results of pulp therapy on certain factors. An analytical study based on radiographic and clinical follow-up examinations. Thesis. 1956;14:1-175.
116. Grahnén H, Hansson L. The prognosis of pulp and root canal therapy. A clinical and radiographic follow-up examination. Odont Revy 1961;12:146-165.
117. Sjögren U, Hägglund B, Sundqvist G, Wing K. Factors affecting the long-term results of endodontic treatment. J Endod 1990;16:498-504.
118. Dalton BC, Ørstavik D, Phillips C, Pettiette M, Trope M. Bacterial reduction with nickel-titanium rotary instrumentation. J Endod 1998;24:763-767.
119. Dakin HD. On the use of certain antiseptic substances in the treatment of infected wounds. Br Med J 1915;ii:318-320.
120. Siqueira JF, Jr., Rôças IN, Favieri A, Lima KC. Chemomechanical reduction of the bacterial population in the root canal after instrumentation and irrigation with 1%, 2.5%, and 5.25% sodium hypochlorite. J Endod 2000;26:331-334.
121. Baumgartner JC, Brown CM, Mader CL, Peters DD, Shulman JD. A scanning electron microscopic evaluation of root canal debridement using saline, sodium hypochlorite, and citric acid. J Endod 1984;10:525-531.
122. Hasselgren G, Olsson B, Cvek M. Effects of calcium hydroxide and sodium hypochlorite on the dissolution of necrotic porcine muscle tissue. J Endod 1988;14:125-127.
123. Clarkson RM, Moule AJ, Podlich H, et al. Dissolution of porcine incisor pulps in sodium hypochlorite solutions of varying compositions and concentrations. Aust Dent J 2006;51:245-251.
124. Moorer WR, Wesselink PR. Factors promoting the tissue dissolving capability of sodium hypochlorite. Int Endod J 1982;15:187-196.
125. Reit C, Dahlén G. Decision making analysis of endodontic treatment strategies in teeth with apical periodontitis. Int Endod J 1988;21:291-299.
126. Ørstavik D, Kerekes K, Molven O. Effects of extensive apical reaming and calcium hydroxide dressing on bacterial infection during treatment of apical periodontitis: a pilot study. Int Endod J 1991;24:1-7.
127. Barbosa CA, Gonçalves RB, Siqueira Jr JF, De Uzeda M. Evaluation of the antibacterial activities of calcium hydroxide, chlorhexidine, and camphorated paramonochlorophenol as intracanal medicament. A clinical and laboratory study. J Endod 1997;23:297-300.
128. Cvek M, Hollender L, Nord CE. Treatment of non-vital permanent incisors with calcium hydroxide. VI. A clinical, microbiological and radiological evaluation of treatment in one sitting of teeth with mature or immature root. Odontol Revy 1976;27:93-108.
129. Katebzadeh N, Hupp J, Trope M. Histological periapical repair after obturation of infected root canals in dogs. J Endod 1999;25:364-368.
130. Leonardo MR, Silveira FF, Silva LA, Tanomaru Filho M, Utrilla LS. Calcium hydroxide root canal dressing. Histopathological evaluation of periapical repair at different time periods. Braz Dent J 2002;13:17-22.
131. Tanomaru Filho M, Leonardo MR, da Silva LA. Effect of irrigating solution and calcium hydroxide root canal dressing on the repair of apical and periapical tissues of teeth with periapical lesion. J Endod 2002;28:295-299.
132. De Rossi A, Silva LA, Leonardo MR, Rocha LB, Rossi MA. Effect of rotary or manual instrumentation, with or without a calcium hydroxide/1% chlorhexidine intracanal dressing, on the healing of experimentally induced chronic periapical lesions. Oral Surg Oral Med Oral Pathol Oral Radiol Endod 2005;99:628-636.
133. Andersen M, Lund A, Andreasen JO, Andreasen FM. In vitro solubility of human pulp tissue in calcium hydroxide and sodium hypochlorite. Endod Dent Traumatol 1992;8:104-108.
134. Türkün M, Cengiz T. The effects of sodium hypochlorite and calcium hydroxide on tissue dissolution and root canal cleanliness. Int Endod J 1997;30:335-342.
135. Wadachi R, Araki K, Suda H. Effect of calcium hydroxide on the dissolution of soft tissue on the root canal wall. J Endod 1998;24:326-330.
136. Teixeira FB, Levin LG, Trope M. Investigation of pH at different dentinal sites after placement of calcium hydroxide dressing by two methods. Oral Surg Oral Med Oral Pathol Oral Radiol Endod 2005;99:511-516.
137. Georgopoulou M, Kontakiotis E, Nakou M. In vitro evaluation of the effectiveness of calcium hydroxide and paramono-chlorophenol on anaerobic bacteria from the root canal. Endod Dent Traumatol 1993;9:249-253.
138. Tronstad L, Andreasen JO, Hasselgren G, Kristerson L, Riis I. pH changes in dental tissues after root canal filling with calcium hydroxide. J Endod 1981;7:17-21.
139. Wang J-D, Hume WR. Diffusion of hydrogen ion and hydroxyl ion from various sources through dentine. Int Endod J 1988;21:17-26.
140. Nerwich A, Figdor D, Messer HH. pH changes in root dentin over a 4-week period following root canal dressing with calcium hydroxide. J Endod 1993;19:302-306.
141. Pérez F, Franchi M, Péli JF. Effect of calcium hydroxide form and placement on root dentine pH. Int Endod J 2001;34:417-423.
142. Nair PNR. Cholesterol as an aetiological agent in endodontic failures—a review. Aust Endod J 1999;25:19-26.
143. Yared GM, Dagher FE. Influence of apical enlargement on bacterial infection during treatment of apical periodontitis. J Endod 1994;20:535-537.
144. Siqueira JF, Jr., Lima KC, Magalhães FA, Lopes HP, de Uzeda M. Mechanical reduction of the bacterial population in the root canal by three instrumentation techniques. J Endod 1999;25:332-335.
145. Rollison S, Barnett F, Stevens RH. Efficacy of bacterial removal from instrumented root canals in vitro related to instrumentation technique and size. Oral Surg Oral Med Oral Pathol Oral Radiol Endod 2002;94:366-371.
146. Coldero LG, McHugh S, MacKenzie D, Saunders WP. Reduction in intracanal bacteria during root canal preparation with and without apical enlargement. Int Endod J 2002;35:437-446.
Address for correspondence/reprints:
Dr David Figdor
517 St Kilda Road
Melbourne, Victoria 3004
Email: [email protected]
*Faculty of Medicine, Dentistry and Health Sciences, School of Dental Science, University of Melbourne and Department of Microbiology, Faculty of Medicine, Nursing and Health Sciences, Monash University.
†Endodontics, Department of Odontology, Faculty of Medicine, Umeå University, Sweden.